
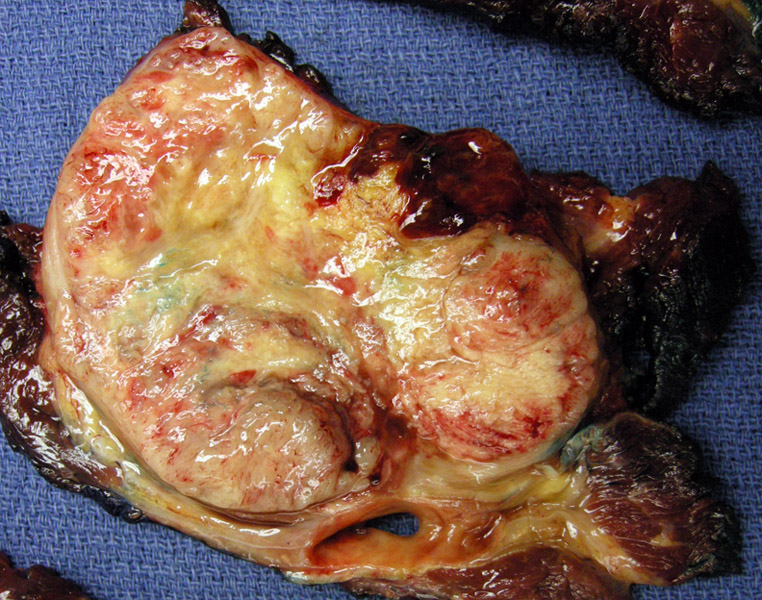
A well -circumscribed extensively necrotic tan lobulated mass is seen measuring 20cm, arising in the thigh. A rim of normal skeletal muscle is seen around the neoplasm.
A cellular area shows cells with eosinophilic cytoplasm growing in a solid sheet. This pattern represents the 'cellular' variant of this entity. Note that these regions bear a strong resemblance to other small blue round cell tumors such as Ewing's sarcoma/primitive neuroectodermal tumors (ES/PNET). More typical areas were also present in this tumor, as shown next.
Malignant cells arranged in loosely anastamosing cords embedded within a myxoid matrix is the characteristic pattern for this tumor.
This image demonstrates both the cellular pattern and the 'classic' pattern. Medium sized blunt polygonal cells with dark nuclei separated by a blue myxoid stroma (on left) with more cellular areas intervening (right). Note that on low power (not demonstrated here), the tumor has a multinodular, lobulated appearance, with the periphery of the lobules more cellular compared to the center.
Extraskeletal myxoid chondrosarcoma (also known as chordoid sarcoma) may have a new name in the future. The current name simply reflects its histology, as extraskeletal mxyoid chondrosarcomas (EMCs) feature chondroblast-like cells set in a myxoid background. However, it is not clear that this soft tissue tumor has true cartilagenous differentiation. Thus, it is listed under a group of tumors with uncertain differentiation in the current WHO classification.
Recent molecular studies have identified a unique balanced translocation t(9;22)(q22:q12) for EMC. This fuses the EWS gene located on 22q12 with the CHN gene (aka NR4A3, NOR1) located on 9q22. The fusion gene product EWS-CHN is found in approximately 70% of EMC, thus though not invariably present in all EMCs, it is pathognomonic when found. Other less common translocation t(9;17)(q22;q11) joins the CHN to the TAF2n gene on 17q11 and is identified in a small percentage of EMCs. A 2001 study by Okamoto of 18 cases of EMC found that the two aforementioned translocations were present in 15 of the 18 cases.1
Immunohistochemical studies have also suggested that EMCs may have neural or neuroendocrine differentiation, as significant proportion of these tumor are positive for NSE (54%), synpatophysin (59%), and PGP9.5 (24%).2 The definition of EMCs will continue to evolve as molecular genetics and other ancillary studies provide increasing information regarding this tumor.
Note that EMCs can rarely occur within the bone, giving the confusing name of "extraskeletal myxoid chondrosarcoma of the bone" or occasionally called just "myxoid chondrosarcoma of the bone" or "mxyoid chondrosarcoma", which then must be differentiated from the conventional chondrosarcoma with myxoid features *sigh*.
Occurs primarily in the deep soft tissue of the proximal extremities and limb girdles, with the thigh being the most common location. It can occur in all ages, however, EMCs is mainly a tumor of adults (peak incidence between the 5th and 6th decades) with a M:F of almost 2:1.2 Presents with a palpable mass with variable pain and tenderness.
There is currently no standard therapeutic protocol. Surgery +/- adjuvant chemotherapy regimens have been employed with varying rates of success. It is hoped that new therapies can be developed to exploit the EWS-CHN gene fusion unique to this tumor.
The 10 year survival rate has been reported to be approximately 70-78%, with high rates of local recurrence after excision (48%) and a tendency to metastasis (46%).2
1 Okamato S et al. Extraskeletal Myxoid Chondrosarcoma: A Clinicaopathologic, Immuhistochemical, and Molecular Analysis of 18 cases. Human Pathology 2001; 32: 1116-1124.
2 Hisaoka M, Hashimoto H. Extraskeletal myxoid chondrosarcoma: Updated clinicopathological and molecular genetic characteristis. Patholgoy International 2005; 55: 453-463.
3 Weiss SW, Goldblum JR. Enzinger. Enzinger and Weiss' Soft Tissue Tumors. 5th Ed. Philadelphia, PA: Elsevier; 2008: 1023-1031.