
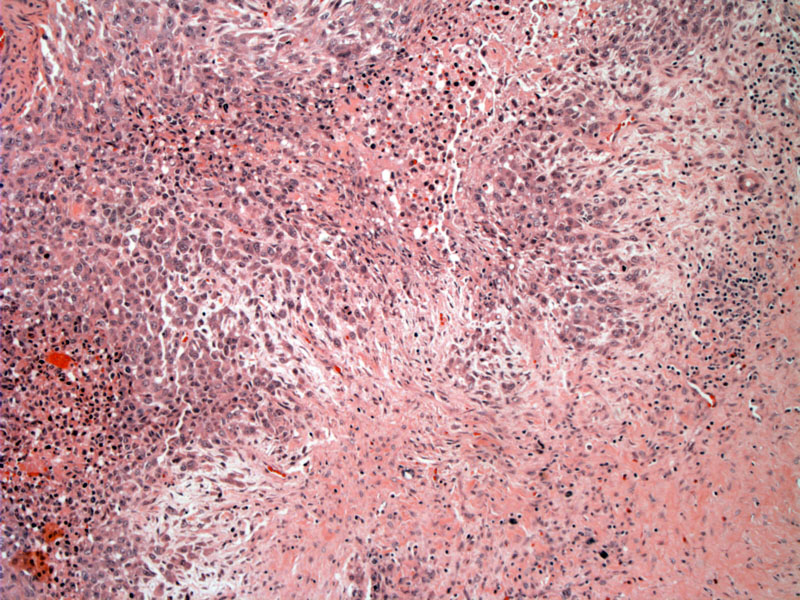
Case 1: There is an undulating border of tumor cells with fibrosis and loose stroma intervening between these irregular sheets. Note that often, the cellular areas cuff the necrotic center to create the characteristic
Higher power image showing cells which lack distinct cohesion to one another but have a distinct epithelioid appearance. Note that some of the cells may have a rhabdoid morphology.
The tumor is composed of plump spindled or epithelioid cells with ample eosinophilic cytoplasm. The nuclei are round with slightly irregular contours, a vesicular chromatin pattern and variably conspicuous nucleoli.
Irregular sheets of cells with intervening stroma is typical.
Spindled, rounded or polygonal tumors cells are intimately blended together with fibromxyoid stroma in this particular area.
Case 2: The nuclei are round, and some have irregular contours. Mild pleomorphism can be present in the distal type.
The tumor cells have a vesicular nuclei with irregularly distributed chromatin pattern. The cells appear to be dissecting through the stroma, taking on an almost spindled look due to this behavior.
Ample eosinophilic cytoplasm is typical of these tumor cells, which have more of a polygonal shape.
The cells in the upper left hand corner may hint at a rhabdoid morphology, but it is not clearly evident. Rhabdoid cells and prominent pleomorphism are much more common in the proximal-type.
The tumor demonstrates strong positivity for EMA.
CAM 5.2 (marker of low molecular weight cytokeratin 8, 18, 19) is focally positive.
CD34 positivity is demonstrated in ~50% of cases, which is helpful in distinguishing this entity from carcinomas (which should be negative for CD34).1
Epithelioid sarcoma has a striking clinical presentation, and often, the diagnosis can be made from history alone. Painful subcutaneous or deep dermal nodules form in the hand or wrist, which eventually ulcerated. These tumors are notorious for spreading along fascial planes, thus, one may see multiple satellite nodules in a line from hand/wrist toward the elbow.1 The tumor's capacity for progressive insidious spread makes local recurrence a common complication, and eventually, 80% of patients die from this tumor after 10-20 years.2
Microscopically, the growth is typically nodular, and the center of the nodule often undergoes degeneration (e.g. necrosis, hemorrhage, cyst formation, calcifications or replacement of tumor cells by a myxohyaline stroma).3 . A proliferation of tumor and inflammatory cells cuff or circle an acellular necrotic center, creating a pseudogranulomatous appearance. This is the most common pattern for this tumor.
A subset of epithelioid sarcomas are located proximally, especially in the pelvis, perineum and vulva. These proximal type epithelioid sarcomas also demonstrate different histology from the conventional type in that the tumor cells are larger, more pleomorphic and tend to be more "rhabdoid" (defined above). These tumor cells grow in diffuse sheets and can resemble a poorly differentiated carcinoma or an extrarenal rhaboid sarcoma. The proximal subtype, also known as "large cell" type, pursues a more aggressive course and undergoes earlier metastasis.
Recent immunohistochemical studies have revealed a consistent loss of INI1 expression in both conventional and proximal-type epithelioid sarcomas. Hornick and colleagues (2009) demonstrated that 127 of 136 (95%) of epithelioid sarcomas showed a loss of INI1 expression.5 Chbani and colleagues (2009) also showed similar results with 86 out of 106 (81%) epithelioid sarcomas demonstrated an absence of INI1 staining.4
INI1, also known as hSNF5, is located on chromosome 22 and functions as a tumor suppressor. Inactivation of this gene is also the hallmark malignant rhabdoid sarcoma, one of the main entities on the differential diagnosis for epithelioid sarcoma. Not surprisingly, both epithelioid sarcoma (especially proximal type) and rhabdoid sarcoma contain a population of cells with rhabdoid morphology.
Epitheliod sarcomas are generally positive for vimentin, keratins, EMA, CD34 and absent for INI1. In a recent study of 106 epithelioid sarcomas, 100% stained for vimentin, 96% for AE1/AE3, 98% for EMA, 66% for CD32, 15% for S-100 and 81% demonstrated loss of INI1 expression.4
Preferentially affects adolescent and young adults, with a male predilection. The most common anatomic site are the distal extremitites, especially the hands and wrist. Proximal locations include the pelvic and inguinal regions (perineum, vulva) and tends to affect an older population (young to middle-age adults).
In terms of the distal type, epithelioid sarcoma is one of the more common soft tissue sarcomas of the upper extremity.
5 year survival is ~50-60%, however, as a result of the tumor's insidious spread, the overall mortality rate that approaches 80% after 10-20 years.2 Metastases to the scalp is particularly common. Prognostic factors include location of the tumor (proximal tumors are more aggressive and metastasize earlier) and tumor size (less than 5 cm is favorable).2
→There is a distal type and a proximal type.
→In the proximal type, the hand is often affected.
→Loss of the INI gene (which acts as a tumor suppressor gene), seen in 80% of epithelioid sarcomas.
→A "pseudogranulomatous" histologic appearance is most common.
→Loss of the INI gene is also seen in malignant rhabdoid tumors, which is on the differential diagnosis for epithelioid sarcoma.
1 Sternberg SS, ed. Diagnostic Surgical Pathology.4th Ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2004: 1829-1835.
2 Fletcher CDM, ed. Diagnostic Histopathology of Tumors. 3rd Ed. Philadelphia, PA: Elsevier; 2007: 1578-1579.
3 Silverberg SG. Silverberg's Principles and Practice of Surgical Pathology and Cytopathology. Philadelphia, PA: Elsevier; 2006: 398-400.
4 Chbani L et al. Epitheliod Sarcoma: A Clinicopathologic and Immunohistochemical Analysis of 106 Cases From the French Sarcoma Group. Am J Clin Pathol 2009;131-222-227.
5 Hornick JL et al. Loss of INI1 Expression is Characteristic of Both Conventional and Proximal-type Epitheliod Sarcoma. Am J Surg Pathol 2009;33:542-550.