
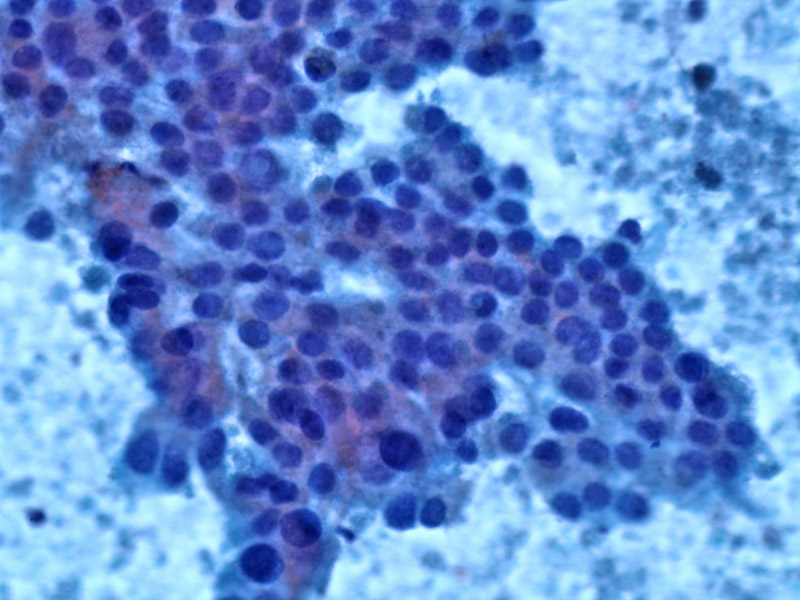
FNA is a sensitive and specific method to diagnosis ductual adenocarcinoma of the pancreas. There is a sheet of epithelial cells with a so-called drunken honeycomb appearance.
The area from the same tumor shows more prominent nuclear pleomorphism, nuclear crowding and apoptotic cells, and tumor necrosis. The range of atypia is far greater than any reactive process could ever account for.
Single malignant cells are dying, in this background of tumor necrosis containing ghosts of cells and debris.
A haphazard arrangement of the neoplastic glands is one of the best diagnostic clues to ductal adenocarcinoma, as this entity can sometimes be difficult to distinguish from reactive glands. This image demonstrates that a single tumor can show a range of atypical nuclei, as both the round gland just above the middle of the field, and those in the lower field, are both part of the same adenocarcinoma.
A residual lobule is seen infiltrated by malignant glands and single cells.
Bizarre malignant cells are seen infiltrating in single cell fashion, no longer bothering to recapitulate glands. Some cells contain a blue-tinged mucin vacuole defining their adenocarcinomatous nature. There is a brisk desmoplastic response to their infiltration.
Ductal adenocarcinoma make up a majority of neoplasms in the pancreas (85-90%), and the majority occur in the pancreatic head (60%), although involvement of the body and tail is not uncommon.
Over 80% of pancreatic adenocarcinomas have a mutation in codon 12 of KRAS and interestingly, the incidence of mutations is higher in smokers versus nonsmokers with the tumor (Hruban).
DPC4 (SMAD4, MADH4) is a tumor suppressor gene and genetic inactivation of this gene is seen in 55% of pancreatic adenocarcinomas. Inactivation of DPC4 is thought to be a critical factor in tumor aquiring the ability to invade and metastasize. Corroborating this assertion is the finding of increased loss of DPC4 in cases of metastasis as well as shortened survival in these patients (Yachida).
Most commonly occurs in older men (M:F ratio is 2:1) with an average age of 60 or older. Risk factors include smoking and a positive family history of pancreatic cancer. Presents as epigastric pain that radiates to the back, often accompanied by weight loss and jaundice.
Surgical resection +/- adjuvant chemotherapy or radiation.
Extremely poor as 80% of patients have metastatic disease at time of presentation. The 5 year survival is less than 2%.1 Specifically, survival curves are significantly worse for those with perineural or vascular invasion histologically, and if more than 20% of lymph nodes were positive. Additionally, a positive resection margin was also associated with shortened survival (Garcea).
• Pancreas : Chronic Pancreatitis
Garcea G. Tumour characteristics predictive of survival following resection for ductal adenocarcinoma of the head of pancreas. Eur J Surg Oncol. 2007 Sep;33(7):892-7.
Hruban RH, et al. K-ras oncogene activation in adenocarcinoma of the human pancreas. A study of 82 carcinomas using a combination of mutant-enriched polymerase chain reaction analysis and allele-specific oligonucleotide hybridization. Am J Pathol. 1993 August; 143(2): 545–554.
Yachida S, Iacobuzio-Donahue CA. The pathology and genetics of metastatic pancreatic cancer. Arch Pathol Lab Med. 2009 Mar;133(3):413-22.
Iacobuzio-Donahue CA, Montgomery EA. Gastrointestinal and Liver Pathology: Foundations in Diagnostic Pathology. Philadelphia, PA: Elsevier; 2005: 473-8.