
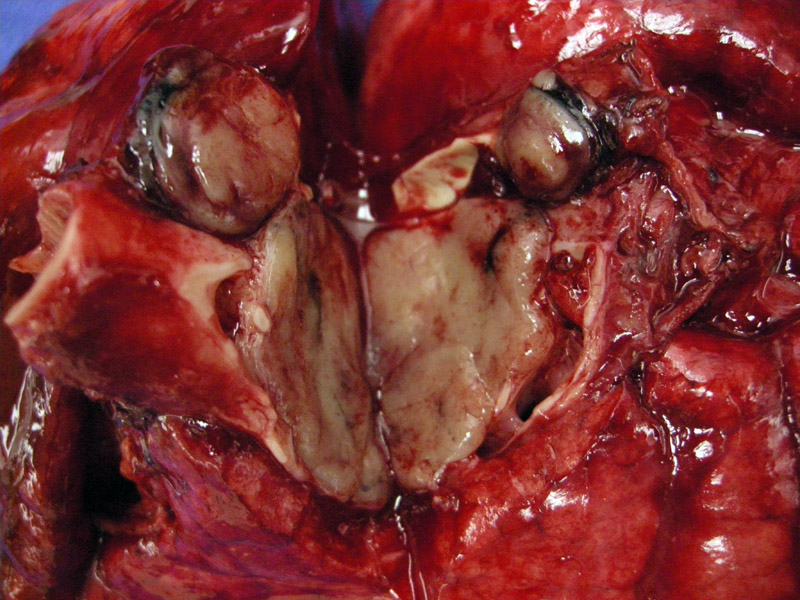
Grossly, typical carcinoid (TC) is an intraluminal solid tan-yellow mass with an overlying epithelium. This explains the common clinical presentation of airway obstruction (wheezing, cough or pneumonia), as the mass can protrude into the airway lumen. TC measure 2 to 4 cm in greatest dimension, and do not demonstrate necrosis or hemorrhage. TC can be central (associated with cartilaginous airways) or peripherally located. In comparison, AC tend to be larger, measuring at least 3 cm in greatest dimension, have a predilection to be peripherally located and may contain foci of necrosis or hemorrhage. Note, however, that in many instances, TC and AC cannot be distinguished by gross or radiographic examination. One must rely on strict histologic criteria (discussed below).
Bronchial epithelium is seen here, thus, the tumor is centrally located and bulging into the bronchial lumen.
Carcinoid can have a range of architectural patterns including nested/insular, trabecular and solid patterns.
The tumor consists of uniform cells with a moderate about of cytoplasm and "salt and pepper" chromatin pattern. The criteria for the diagnosis of typical carcinoid are (1) less than 2 mitoses per 10 HPF (or 2 mm2 ) (2) lack necrosis and (3) 0.5 cm or larger. Tumors smaller than 0.5cm are referred to as a tumorlets. In comparison, the criteria for the diagnosis of atypical carcinoid is 2-10 mitoses per 10 HPF OR presence of punctate necrosis.2
Usually, carcinoid has a delicate fibrovascular background, but sometimes, dense sclerosis can occur in the stroma.
Note again the cartilege (on the right) and the nested configuration of the tumor. This tumor is located centrally. As an interesting sidenote, typical carcinoid located in the periphery (not associated with cartilege) can have a spindled morphology not usually found in those centrally located or in other neuroendocrine tumors.1
The terminology and classification of neuroendocrine tumors of the lung is currently evolving. The terms used in the current WHO classification to describe tumors with NE morphology are: carcinoid tumor, atypical carcinoid/well-differentiated NE carcinoma, large cell neuroendocrine carcinoma and small cell carcinoma. Also used are the descriptors well-differentiated (for atypical carcinoids), moderately differentiated (for large cell NE carcinoma) and poorly-differentiated (for small cell carcinoma).
These terms have caused quite a bit of confusion among pathologists and clinicans. For example, the term carcinoid is often associated with a benign tumor in the minds of some clinicans, however, ALL carcinoid tumors have malignant potential. Therefore, the terms typical carcinoid and atypical carcinoid may give a false impression that these tumors cannot metastasize. Furthermore, referring to LCNC as moderately differentiated does not accurately portray the aggressiveness of this tumor.1
A new scheme has been proposed to reflect the malignant potential of these tumors and to better stratify the tumors by their biologic behavior. Grade I neuroendocrine carcinoma (formerly classic or typical carcinoid), Grade II neuroendocrine carcinoma (atypical carcinoid), and Grade III neuroendocrine carcinoma (LCNC, SCLC or a combination of LCNC/SCLC with another histologic type).1
For the purposes of this discussion, the classification described by Travis (2006) is utilized, as it is simple and also accurately describes the biologic features of the tumors. The four entities are thus: low-grade typical carcinoid (TC), intermediate-grade atypical carcinoid (AC), high-grade large cell NE carcinoma (LCNC) and high-grade small cell lung carcinoma (SCLC).
When signing out a case using any of the above classification schemes, one might consider adding a brief discussion of the terminology and stressing that all carcinoids have malignant potential.
TC, AC, LCNEC and SCLC are neuroendocrine tumors with shared morphologic, immunohistochemical and molecular features and are often considered different entities along a spectrum. However, it might be best to consider TC and AC as a group; and LCNEC and SCLC as a separate group, as these two groups are very different from each other clinically and microscopically.
For example, TC/AC present in younger patients (TC at 45-50 years, AT at 56-63 years versus SCLC and LCNEC at 64-67 years). Virtually all SCLC and LCNEC patients have a smoking history, whereas 20-40% of TC/AC patients are nonsmokers. TC/AC can occur in association with MEN type I and LCNEC/SCLC do not have this association. Neuroendocrine cell hyperplasia and tumorlets are often seen adjacent to TC/AC and are considered by some authorities to be pre-neoplastic lesions to TC/AC, whereas these entities are not seen in LCNEC/SCLC. Furthermore, LCNEC/SCLC are often seen in combination with other histologic types such as squamous cell carcinoma or adenocarcinoma, whereas TC/AC are not usually combined with other tumor types.2
Carcinoid tumors should most often be treated by lobectomy with lymph-node dissection as a standard procedure
One large study found a 5-year survival of nearly 90% in patients with localized disease and 70% in patients with regionally advanced or metastatic disease, which reflects data from other large studies as well. However, atypical carcinoids more often present at a more advanced stage, are more likely to recur regardless of stage, and are associated with a decreased 5-year survival compared to typical carcinoid tumors.3 In contrast, SCLC and LCNEC have very poor outcomes and few patients experience long term survival.
1 Cerilli LA et al. Neuroendocrine Neoplasms of the Lung. Am J Clin Pathol 2001;116 (Suppl 1):S65-S96.
2 Travis WD. Neuroendocrine Lung Tumors. Pathology Case Reviews (2006)11:2350242.
3 Ferguson MK, et al. Long-term outcome after resection for bronchial carcinoid tumors. Eur J Cardiothorac Surg 2000;18:156-161.
For a more comprehensive understanding of neuroendocrine tumors of the lung, please visit our cases describing atypical carcinoid, small cell carcinoma.