
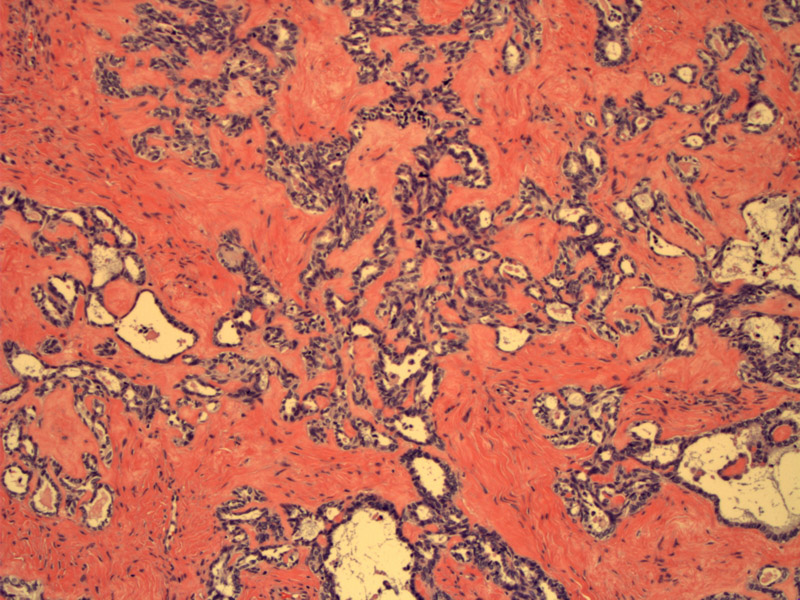
Infiltrative tubules of various sizes are seen in a hyalinized desmoplastic stroma. This pattern is most suggestive of the tubular variant of mesonephric adenocarcinoma (which would be found in the cervix). Normal ovarian stroma is not seen.
Note that the glandular lumens contain eosinophilic secretions, a helpful diagnostic feature, as these secretions are found in mesonephric rests, mesonephric hyperplasia and mesonephric adenocarcinomas. The cells lining these tubules exhibit moderate cytologic atypia (high N/C ratio, hyperchromasia and pleomorphism).
Note the irregular ragged outlines of the glands. The glands are also focally crowded (back-to-back glands). In contrast, benign mesonephric lesions are lined with bland cuboidal epithelium and the tubules have a rounded coutour.
Mesonephric adenocarcinoma is an uncommon malignancy derived from mesonephric remnants (mesonephric rests) in the cervix. This tumor exhibits a wide range of histological patterns including ductal, tubular, retiform, solid, sex-cord like and spindled. The ductal and tubular patterns are most common, resembling endometrioid adenocarcinma and mesonephric hyperplasia, respectively. Often, a mixture of patterns is seen. The spindled pattern is a biphasic tumor consisting of a spindled component (resembling endometrial stromal sarcoma) blending with the glandular component, often termed 'malignant mixed mesonephric tumor'.
Due to the rarity of this tumor, prognostic factors have not been clearly delineated. However, stage is important. Furthermore, a spindled component ('malignant mixed mesonephric tumor') may have a poorer prognosis.
Most mesonephric adenocarcinomas have a fairly indolent course and present at an early stage. However, there is clear documentation in the literature of recurrent and metastatic disease, as in this particular case. Prognosis is poor for these patients.
• Cervix : Mesonephric Adenocarcinoma