
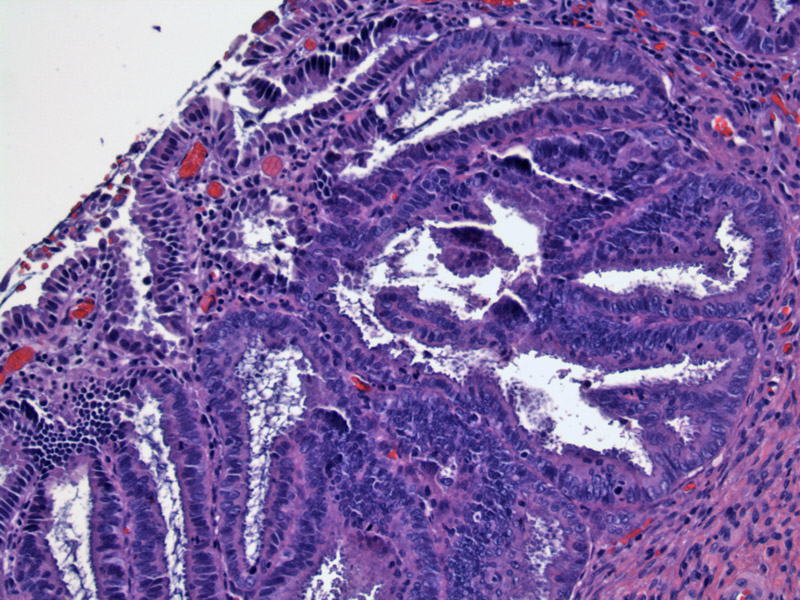
Glandular architecture is usually preserved, but glands may be crowded and exhibit increasing complexity such as papillary infoldings and cribriforming. The cells lining the glands are crowded, enlarged, hyperchromatic, stratified or pseudostratified. Nucleoli are usually inconspicuous but may be prominent. Mitotic figures (essential to the diagnosis) are usually located toward the luminal side of glands.
This case demonstrates the intestinal subtype, with prominent goblet cells. There are several histologic subtypes of ACIS. Although there is no difference in prognosis or tumor behavior among the various subtypes, they are important to recognize as the morphology of ACIS can be quite diverse.
Brisk mitotic activity must be present to diagnose ACIS. Mitotic figures are often located toward the luminal side of the gland and appear
Cytologically, clusters of hyperchromatic cells are seen. The nuclei have granular chromatin with inconspicuous nucleoli, although in this image, the nucleoli are prominent in some of the cells, which is also normal in ACIS.
Some of the cells demonstrate elongated nuclei. Size variation can be mild or moderate, but always with an increased N/C.
Another cell cluster demonstrates elongated nuclei, some of them located at the periphery give the cell cluster a feathered-edge appearance.
A different patient with ACIS shows a hyperchromatic small cluster of endocervical cells.
Other areas show elongated nuclei with hyperchromasia and crowding.
Adenocarcinoma in situ (ACIS) is the established precursor to most forms of cervical adenocarcinoma. ACIS have been observed to progress to invasive adenocarcinoma and is commonly found adjacent to early invasive foci of adenocarcinoma. ACIS is thought to arise from the basal/reserve cells located in the squamocolumnar junction that have the capacity to differentiate into the squamous or columnar cells.
The most cases, the lesion originates at the squamocolumnar junction and is continguous from that point. However, ACIS occasionally may be multifocal or be situated proximal (higher) to the endocervical canal. It may involve the lower uterine segment and endometrial cavity. Approximately 50% of ACIS is associated with a squamous intraepithelial lesion and >90% have detectable high-risk HPV types (primarily type 16 and 18)(Fletcher).
Several subtypes of ACIS exist, and it is important to recognize that this lesion can have a diverse morphology. The endocervical subtype is most common, which resembles normal endocervical glands with superimposed dysplastic change. The endometrioid subtype resembles well-differentiated neoplastic endometrial glands -- the intestinal subtype exhibits less hyperchromasia and crowding, but is easy to recognize by the presence of goblet cells.
Tuboendometrial metaplasia (TEM), a benign change of endocervical glands, may exhibit crowding, hyperchromasia and nuclear enlargement. Thus, TEM must often be distinguished from ACIS. Mitotic figures are not found in TEM, and TEM is generally located in the upper cervical canal, whereas ACIS is typically located in the squamocolumnar junction. Furthermore, cells in TEM are often ciliated (recapitulating cells that line the fallopian tube). IHC panels will often help to separate the two. MIB-1 (an antibody against ki-67) is a marker of proliferative activity is often positive in ACIS, but negative in TEM. Vimentin also stains strongly in TEM, but not ACIS (Sternberg).
Presents at an average of age of 38, approximately 10 years earlier than invasive adenocarcinoma. Usually asymptomatic, but may present with abnormal vaginal bleeding. Since many women may still be in their reproductive years, treatment considerations include cervical conization (with close followup) rather than hysterectomy.
Note that Pap smears are less sensitive in detecting ACIS and invasive adenocarcinoma because there are many benign conditions that mimic the cytology of atypical glandular cells such as cervicitis, Arias-Stella reaction and tubal metaplasia. Furthermore, endocervical glands are further up the endocervical canal and thus, harder to sample thoroughly.
Thus, the category of AGUS “atypical glandular cells of undetermined significance” on cytology has been a source of much controversy in part due to sampling issues and interobserver variability. Although AGUS is reported in a small percentage (~1%) of all Pap smears, these cases often harbor a significant percentage of cervical neoplasia (Covell).
Usually discovered on cervical biopsy, treatment is best performed with a cervical conization procedure, preferably a cold knife cone biopsy as there is a high prevalence of concurrent invasive adenocarcinoma in women with a diagnosis of adenocarcinoma in situ. Definitive treatment can be performed via simple hysterectomy though this is less favored the patient may need more aggressive therapy with a uterus in situ if concurrent invasive adenocarcinoma is discovered.
Studies have found that patients with positive margins on conization have a significant likelihood of having residual or recurrent disease (60-70%), but management of those with negative margins remains controversial (Salani, Young). This is because even with negative conization margins, there remains a risk of residual, recurrent, or invasive disease (Young). For example, in one large study, 13% of patients with negative margins on conization were found to have residual ACIS or recurrent disease and 2 patients had a diagnosis of invasive adenocarcinoma during follow-up. Overall, pooling of data from multiple studies examining those with negative margins yields a 9% rate of recurrence (Young).
→ACIS may be reported as "atypical glandular cells of undetermined significance" on cytology.
→ACIS is often found in adjacent to invasive adenocarcinoma and 50% of ACIS may be associated with a squamous intraepithelial lesion.
→>90% of ACIS are associated with a high-risk HPV type (primarily type 16 and 18).
• Cervix : Adenocarcinoma, Villoglandular Type
Covell JL. Atypical Glandular Cells and Endocervical Adenocarcinoma in Situ of the Uterine Cervix. Pathology Case Reviews. 2005;10:155-164.
Fletcher CDM, ed. Diagnostic Histopathology of Tumors. 3rd Ed. Philadelphia, PA: Elsevier; 2007: 699-701.
Salani R, Puri I, Bristow RE. Adenocarcinoma in situ of the uterine cervix: a metaanalysis of 1278 patients evaluating the predictive value of conization margin status. Am J Obstet Gynecol. 2009 Feb;200(2):182.e1-5.
Sternberg SS, ed. Diagnostic Surgical Pathology.4th Ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2004: 2411-2414.
Young JL, et al. Cervical adenocarcinoma in situ: the predictive value of conization margin status. Am J Obstet Gynecol. 2007 Aug;197(2):195.e1-7; discussion 195.e7-8.