
An overall papillary architectural pattern is very common, with small glands proliferating within the papillae. Although most glands are lined by columnar or cuboidal cells, the glandular lining can also be attenuated. A foci of squamous metaplasia can be seen in the top center image.
A closer view demonstrates glands lined with flattened epithelium. There are also numerous vacuolated cells packed between the glands and a sparse inconspicuous stroma with scattered inflammatory cells. Intracytoplasmic mucin vacuoles are evident (stains light blue).
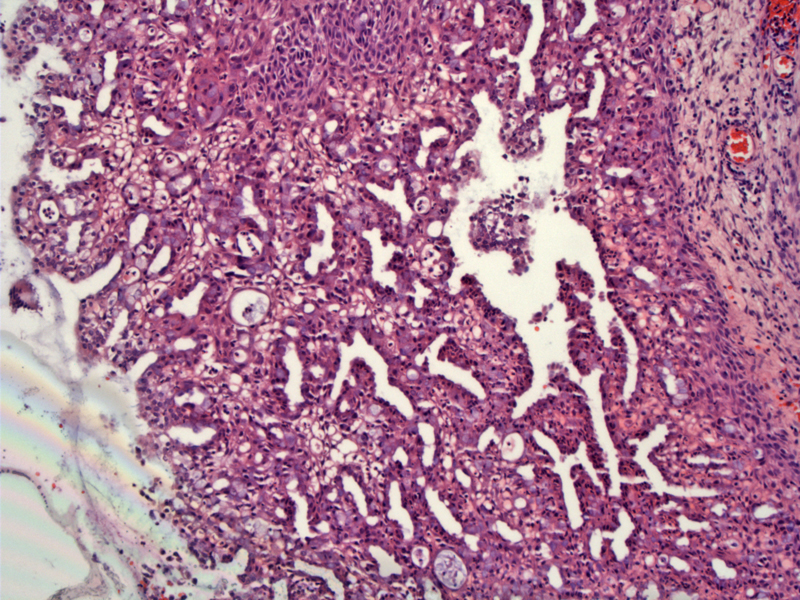
In this different case of microglandular hyperplasia, the columnar cells have 'matured' into metaplastic squamous epithelium. A papillary architecture can be appreciated.
The glandular lumen contains mucin and neutrophils and scattered inflammatory is seen in the stroma.
Another image of proliferation of small glands along with some squamous metaplasia. Inflammatory cells are seen within the glandular lumens as well as in the stroma.
Microglandular hyperplasia, also known as microglandular adenosis, was originally thought to be related to the use of oral contraceptives, however, this causal relationship has been questioned since these changes can be seen in women without a history of OCP use and in post-menopausal women.1
Microscopically, there is a spectrum of changes that begins with columnar cell proliferation and terminates in squamous metaplasia.2 Columnar cell proliferation is characterized by a dense proliferation of small glands lined by a single layer of columnar or cuboidal cells. Subnuclear and supranuclear vacuoles are highly characteristic of these glandular cells. The nuclear features should be bland with little atypia or hyperchromasia. Mitotic activity is minimal. Mucin secretions and neutrophils can be seen within glandular lumens. There is invariably scattered inflammatory cells throughout the stroma. In later stages, the columnar epithelium is gradually replaced by metaplastic squamous epithelium.2,3
One of the main differential diagnosis for microglandular hyperplasia is clear cell carcinoma of the cervix, especially if the glands form a solid pattern. Features that help distinguish between the two entities include (1) presence of subnuclear vacuoles and intracytoplasmic mucin in the glandular cells in microglandular hyperplasia (2) only focal and minimal atypia and rare mitotic figures in microglandular hyperplasia (3) lack of glycogen in glandular cells versus abundant glycogen in clear cell carcinoma (4) presence of reserve cell hyperplasia and squamous metaplasia in microglandular hyperplasia.4 Remember that basal (reserve) cells reside in endocervical glands in the squamo-columnar junction that can differentiate into endocervical glandular cells or squamous cells.
Lastly, CEA is positive in endocervical neoplasia (including clear cell adenocarcinoma) and negative in microglandular adenosis.
Benign entity; however, microglandular adenosis can form a polypoid lesion with complex histologic features, thus, it must differentiated from clear cell carcinoma and mucinous adenocarcionma of the cervix. A key feature is that microglandular hyperplasia lacks cytologic atypia and mitotic activity.
• Cervix : Clear Cell (Adeno)carcinoma
1 Kumar V, Abbas AK, Fausto N. Robbins and Cotran Pathologic Basis of Disease. 7th Ed. Philadelphia, PA: Elsevier; 2005: pg 1527-8.
2 Sternberg SS, ed. Diagnostic Surgical Pathology.4th Ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2004: 2405-6.
3 Kurman RJ, Norris HJ, Wilkinson E. Tumors of the Cervix, Vagina and Vulva: Atlas of Tumor Pathology. Third Series Fascicle 4. Washington DC; AFIP: 1990: 119-120.
4 Fletcher CDM, ed. Diagnostic Histopathology of Tumors. 3rd Ed. Philadelphia, PA: Elsevier; 2007: 721.