
Aggressive (deep) angiomyxoma is a paucicellular neoplasm consisting of spindle-shaped cells with delicate cytoplasmic processes embedded in a loose myxoid stroma. Medium to large vessels, often thick-walled and hyalinized, are haphazardly arranged throughout the neoplasm.
The vessel walls have a characteristic tendency to be thickened and hyalinized. Often, delicate fibers of collagen and smooth muscle will condense around the vessels.
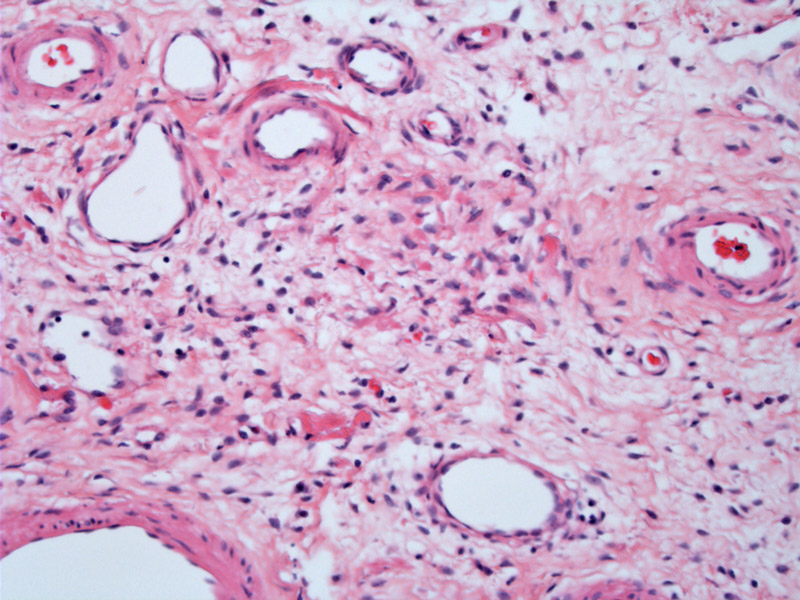
Although most blood vessels are medium to large vessels, there is a wide range of vessel size. As seen in this image, smaller vessels are also present. Note the prominent wall of the central large vessel.
Note the stellate and spindled cells with delicate cytoplasmic processes in loose myxoid stroma. The spindled cells are monomorphic and without atypia. Mitotic figures are not present. The overall look is that of a hypocellularity with copious myxoid stroma.
The tumor is unencapsulated and infiltrates into surrounding connective tissue, blending imperceptibly with non-neoplastic mesenchyme. It is this feature that often leads to incomplete excision and subsequent recurrence.
Deep (aggressive) angiomyxoma typically occurs in the pelvis and perineum of women, although some cases have been reported in the inguinoscrotal region of men. A characteristic feature of this tumor is its tendency to infiltrate insidiously into surrounding tissues. It may be difficult to distinguish neoplastic tumor from non-neoplastic tissue, thus clear surgical margins may be difficult to obtain. Recurrence is common after initial excision, sometimes occurring decades after the initial surgery.
Because deep angiomyxomas invade locally and recur, it is important to distinguish this tumor from other benign stromal tumors of the vulva, which do not recur. These other benign entities include fibroepithelial stromal polyp, angiomyofibroblastoma and cellular angiofibroma.
Recent studies have revealed that the most common chromosomal abnormality in aggressive angiomyxoma is rearrangement of 12q15, where HMGA2 (nuclear transcription factor) is located. Up to 30% of AA show this rearrangement. Note, however, that immunoreactivity for HMGA2 (seen in 50% of AA) does not necessary correlate with the presence of the gene rearrangement (Oliva).
HMGA2 positivity is also aberrantly expressed in occasional vulvovaginal smooth muscle neoplasms -- it has emerged as a helpful stain in distinguishing aggressive angiomyxomas from other stromal neoplasms and in some cases, assessing resection margins (Oliva, McCluggage 2009).
Differential diagnosis (Oliva):
Fibroepithelial stromal polyps are polypoid with a circumscribed border. The vascular component is located at the core of the polyp. In contrast, AA has an infiltrative border and the vascular component is haphazardly distributed. The polyp can twist and cause edema in the stroma, but the stroma is not truly mxyoid as in AA. Fibroepithelial polyps also have characteristic multinucleated stromal cells just beneath the epithelial surface; these cells are not present in AA.
Angiomyofibroblastoma is subcutaneous well-circumscribed nodule with a vascular component consisting of small capillary sized vessels. In contrast AA is a deep-seated lesion with infilrative borders and medium-sized vessels with hyalinized walls. There is alternating zones of cellularity whereas AA is largely paucicellular.
Superficial angiomyxoma is also a mxyoid neoplasm with bland spindle cells. However, it occurs in a cutaneous or subcutaneous location with more delicate vessels. Often, adnexal structures indicating its superficial location. IHC can also be helpful as the spindle cells are negative for desmin, whereas the spindle cells in AA are positive for desmin.
Occurs most often in the 4th decade. Presents as a non-painful cyst-like lesion or ill-defined swelling of the vulvar region. Initial clinical impression is often that of a Bartholin's gland cyst. The tumor is often greater than 10 cm at the time of diagnosis.
Surgical excision with wide margins whenever possible.
Interestingly, McCluggage and colleagues (2006) described a case of recurrent aggressive angiomyxoma where GnRH agonist administration lead to the involution of the tumor. The premise behind this therapy is based on presence of estrogen and progesterone receptors in these tumors, and the stromal cells are considered to be specialized genital fibroblasts.
Although GnRH agonists initially stimulates the pituitary into producing FSH and LH, constant administration leads to desensitization and there is a decrease in FSH and LH, thus creating a hypoestrogenic state. Although further investigation is necessary, GnRH agonists may be of value, for either primary or recurrent, which are not amenable to surgical excision. Alternatively, these agents may also be used to reduce tumor volume, so that more conservative surgery can be performed.
The authors do not know if withdrawal of the GnRH agonist will lead to regrowth of the tumor in this patient, as rapid regrowth of leiomyomas of the uterus treated via GnRH agonist is common if the drug is discontinued. Continuous administration of GnRH leads to osteoporosis, thus, alternative therapies that block endogenous estrogen such as aromatase inhibitors may be worth exploring in the treatment of this rare and locally aggressive tumor.
Usually slow growing and locally infiltrative, extending insidiously into adjacent soft tissues, but does not metastasize. Up to 30-40% of cases recur, most likely as a result of incomplete excision. Wide excision with 1 cm margins is recommended. More than one recurrence is rare (Nucci, Fletcher). There have only been two documented reports of metastasis (lungs) and both patients died of the disease.
• Paratesticular : Aggressive Angiomyxoma
• Vulva : Cellular Angiofibroma
• Vulva : Angiomyofibroblastoma
• Vulva : Superficial Angiomyxoma
• Vulva : Superficial Myofibroblastoma
• Vulva : Fibroepithelial Stromal Polyp
Balzer BL, Longacre TA. Aggressive Angiomyxoma of the Femal Genital Tract. Pathology Case Reviews. 2005;10: 46-54.
Fletcher CDM, ed. Diagnostic Histopathology of Tumors. 3rd Ed. Philadelphia, PA: Elsevier; 2007: 742-3.
McCluggage WG, Jamieson T, Dobbs SP, Grey A. Aggressive angiomyxoma of the vulva: Dramatic response to gonadotropin-releasing hormone agonist therapy. Gynecol Oncol. 2006 Mar;100(3):623-5.
McCluggage WG. Recent developments in vulvovaginal pathology. Histopathology. 2009 Jan;54(2):156-73.
Nucci MR, Oliva Esther. Gynecologic Pathology: Foundations in Diagnostic Pathology. Philadelphia, PA: Elsevier: 2009: p34-5.
Oliva Esther ed. Current Concepts in Gynecologic Pathology: Mesenchymal Tumors of the Female Genital Tract: Surgical Pathology Clinics. Philadelphia, PA: Elsevier: December 2009, Vol 2, No 4; p 603-6.