
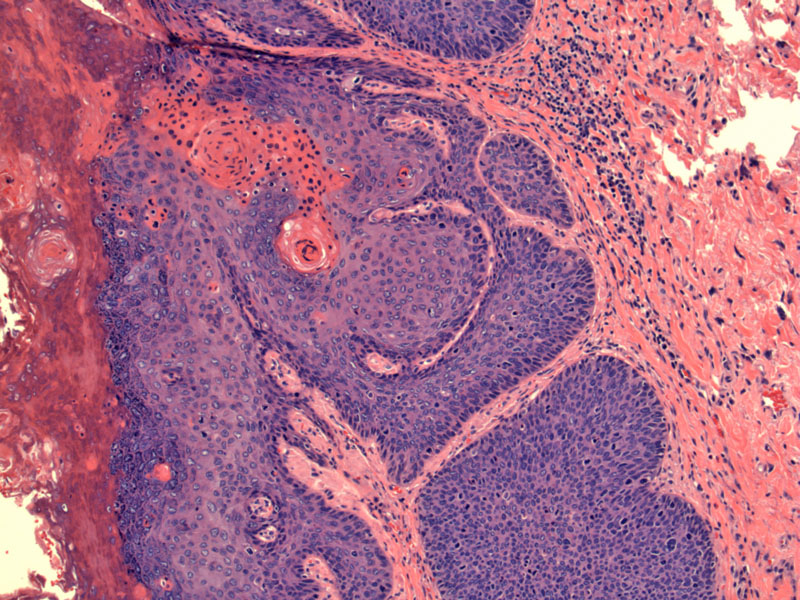
Full thickness dysplasia can be appreciated with pleomorphic enlarged nuclei and brisk mitotic activity.
Anal intraepithelial neoplasia is strikingly similar to cervical intraepithelial neoplasia. Note the immature squamous cells are present throughout the full thickness of the epithelium, and are covered by a parakeratotic layer.
Apoptotic cells are very common.
Squamous cell carcinoma of the anal canal shares many clinical and pathologic similarities with squamous cell carcinoma of the cervix. For example, HPV infection (especially types 16 and 18) is implicated in both conditions and both arise in the transitional zone, which is the squamocolumnar junction in the cervix and bordering the dentate line (anorectal junction) in the anal canal.
In the dentate line (a.k.a pectinate line), simple columnar epithelium with goblet cells (resembling the colon) abruptly transitions to a stratified squamous epithelium. Again, similar to CIN, AIN (anal intraepithelial neoplasia) can be graded from I (mild dysplasia) to III (high grade dysplasia). Koilocytic change indicative of HPV effect can sometimes be seen in the superficial epithelial layers, especially in perianal lesions.
Condylomata and AIN show abnormal Ki-67 staining extending into the upper two-thirds of the epithelial thickness, but positive p16/CDKN2A stain is only present in high-grade AIN (Pirog). Among pathologists, the diagnoses of AINII and AINIII are highly reproducible, but there is a trend to overdiagnose anal condylomata and AIN1 (Pirog).
Note that squamous cell dysplasia or carcinoma of the anus can either refer to inside the anal canal (anal intraepithelial neoplasia) or the perianal skin (Bowen disease). You ought to make this distinction because the epidemiology between these two locations are different. For example, AIN is associated with homosexual men and HIV infection. Bowen disease of the perianal skin is twice as common in women (Iacaobuzio).
AIN and anal SCC usually begins at or above the dentate line and extends upward, and thus, usually presents as an ulcerative lesion of the lower rectum (Fletcher). SCC of the anal canal can be large cell (with or without keratinizing features) or basaloid (formerly termed cloacogenic carcinoma).
There are no randomized controlled trials of treatments to allow for definite treatment recommendations. In general, treatment falls into three categories: local treatment with clinician or patient-applied creams and liquids, clinician-applied ablative techniques such as electrocautery, carbon dioxide (CO2) laser, and/or infrared coagulation.
If left untreated, high grade AIN potentially may progress, especially in immunosuppressed individuals with extensive disease. In HIV-positive individuals, restoration of immune function with highly active antiretroviral therapy (HAART) has not demonstrated substantial benefit in reducing incidence of HG AIN or regression of existing high-grade lesions (Park). Interestingly, several studies have demonstrated a higher incidence of anal cancer since the introduction of HAART, possibly attributable to increased overall survival, allowing HIV-positive individuals to live with oncogenic HPV infection long enough to develop anal cancer.
• Anal : Squamous cell carcinoma, Basaloid type
Iacobuzio-Donahue CA, Montgomery EA. Gastrointestinal and Liver Pathology: Foundations in Diagnostic Pathology. Philadelphia, PA: Elsevier; 2005: 411-4.
Fletcher CDM, ed. Diagnostic Histopathology of Tumors. 3rd Ed. Philadelphia, PA: Elsevier; 2007: 409-410.
Pirog EC, Quint KD, Yantiss RK. P16/CDKN2A and Ki-67 enhance the detection of anal intraepithelial neoplasia and condyloma and correlate with human papillomavirus detection by polymerase chain reaction. Am J Surg Pathol. 2010 Oct;34(10):1449-55.
Park IU, Palefsky JM. Evaluation and Management of Anal Intraepithelial Neoplasia in HIV-Negative and HIV-Positive Men Who Have Sex with Men. Curr Infect Dis Rep. 2010 Mar;12(2):126-133.