
This low power view illustrates some of the features of spindle cell leiomyosarcoma: hypercellularity, prominent nuclear atypia, and high mitotic rate generally exceeding 10 mitotic figures per 10 high-power-fields.
A mitotic figure is seen in the upper center image.
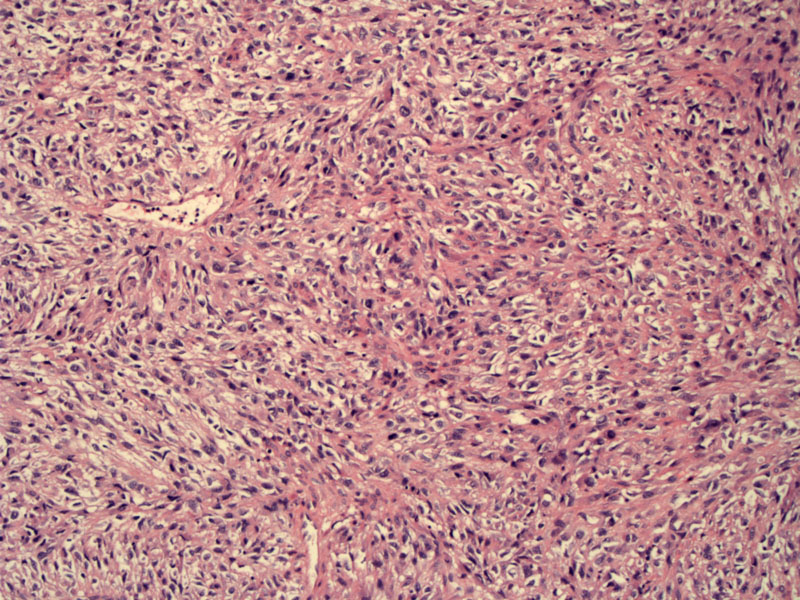
Spindle cell leiomyosarcoma consists of spindle cells with hyperchromatic nuclei and eosinophilic fibrillary cytoplasm.
A fascicular growth of spindled cells exhibit moderate atypia is seen. The lesion is quite cellular.
Coagulative necrosis demonstrates an abrupt transition between viable and dead cells.
Two variably differentiated areas are seen here: a well-differentiated and relatively bland spindle cell proliferation (bottom) abruptly transitions to another spindle cell area with higher grade features. In cases in which only the well-differentiated areas comprise a tumor, the question of the minimal pathological criteria for the diagnosis of leiomyosarcoma becomes problematic. The differential diagnosis has to be reported, which includes smooth muscle tumors of uncertain maligant potential (STUMP).
Again, two areas are seen in this spindle cell leiomyosarcoma. A paucicellular area transitioning to a hypercellular area. Nuclear pleomorphism is prominent.
In this epithelioid variant, round to oval cells with bizarre nuclei are arranged in diffuse sheets. Note the presence of a small bundle of myometrium (top left corner). The rest of the myometrium is overrun by tumor.
Pleomorphic epithelioid cells show widespread invasion into the myometrium (seen at left).
Rarely such tumors may appear myxoid, as in this variant demonstrating myxoid acellular material dissecting between malignant tumor cells.
Uterine leiomyosarcoma is the most common uterine sarcoma, comprising 25-40% of all malignant mesenchymal tumors of the organ, however, it is a rare tumor overall and represent less than 2% of all uterine malignancies. Unlike leiomyomas which demonstrate aberrations in the HMGA2 and HMGA1 gene in a significant percentage of tumors, leiomyosarcomas exhibit a wide range of genetic abnormalities with no particular pattern, indicating a high degree of genomic instability (Fletcher, Nucci).
Grossly, the tumor (mean diameter of 10 cm) is usually located in the submucosa or myometrium. A serosal locaction is uncommon. 5% of cases can arise in the cervix. Leiomyosarcomas are less well-circumscribed than leiomyomas and cannot be easily 'shelled out'. Obvious myometrial invasion may be evident. The cut surface is also softer than leiomyomas and there may be foci of hemorrhage and necrosis.
Leiomyosarcomas come in three main flavors: spindle cell, epithelioid and myxoid. Each of these subtypes has distinctive diagnostic criteria (Fletcher, Nucci, Mills).
Spindle cell is the most common type and resembles a benign leiomyoma, composed of tightly interwoven fascicles of spindled cells. Infiltration into surrounding myometrium may be evident. The lesional cells have elongated blunt-ended nuclei (cigar-shaped) nuclei with eosinophilic fibrillary cytoplasm. The tumor must exhibit two of the following three features to be diagnosed as a leiomyomsarcoma: (1) diffuse moderate or severe nuclear atypia; (2) >10 mitotic figures per 10 HPF; (3) tumor cell necrosis.
Tumor cell necrosis which establishes the diagnosis of leiomyosarcomas must be of the coagulative type. Features of coagulative necrosis include: (1) an abrupt transition between necrotic and viable cells, as opposed to a transition zone of granulation tissue or hyalinized tissue seen in ischemic necrosis; (2) preserved nuclei with marked pleomorphism (atypical ghost nuclei) within necrotic areas; (3) apoptotic cells and nuclear debris at the interface; (4) an irregular, angulated border between necrotic and viable cells, so called 'geographic necrosis'. A cuff of viable and usually pleomorphic cells may be seen around blood vessels.
Coagulative necrosis must be distinguished from 'hyalinizing', or 'bland' necrosis, which is a common finding in benign leiomyomas and mostly due to infarction. Briefly, hyalinizing necrosis exhibits a zone of eosinophilic hypocellular collagen at the interface of the dead and viable cells. This collagen matrix is seen rather than apoptotic cells and necrotic debris found in coagulative necrosis. Dead nuclei may occasionally be seen in areas of hyalinization, however, the nuclei are uniform and faint, whereas the nuclei of ghost cells in coagulative necrosis are more abundant, pleomorphic and hyperchromatic.
Epitheliod leiomyosarcoma: If >50% of cells have an epithelioid appearance (rounded tumor cells with abundant eosinophilic or clear cytoplasm), the tumor is classified as an epithelioid leiomyosarcoma. The epithelioid cells are most often arranged in a diffuse sheet, however, nest, cords and pseudoglandular formation can also be seen. Because of the rarity of this variant, the diagnostic criteria has not been clearly established. As a general guideline, if the following features are found: >3 mitoses/10 HPF, diffuse moderate or several cytologic atypia, tumor necrosis or vascular invasion, infiltrative margins -- consider the diagnosis of epithelioid leiomyosarcoma.
Myxoid leiomyosarcoma: The cut surface of this variant is usually gelatinous due to the myxoid content. Microcopically, spindled or stellate tumor cells are set in a abundant pale blue-grey, extracellular myxoid matrix. The cells should exhibit at least focal moderate to marked cytologic atypia, however, large areas of the tumor may be cytologically bland. Similar to epitheliod leiomyosarcoma, this variant is quite rare and diagnostic criteria has not been clearly established. However, the identification of the following features should prompt one to consider the diagnosis: (1) moderate to marked cytologic atypia; (2) coagulative tumor necrosis; (3) greater than 2 MF/10 HPF; (4) infiltration of surrounding myometrium. Because myxoid tumors can be bland and it only takes two mitotic figures in 10 high power fields to establish the diagnosis, thus extensive and exhaustive sampling must be performed on a myxoid smooth muscle tumor.
Age at diagnosis is between the 5th and 6th decades, approximately 10 years older than women with benign leiomyomas. Incidence appears to be higher in young black women, although not as high as the incidence of leiomyomas in this population. Most common presentation is vaginal bleeding and pelvic pain. Exam usually reveals an enlarged uterus. However, in a postmenopausal woman with rapidly enlarging 'fibroid' should raise the possibility of leiomyosarcoma. If the neoplasm does not involve the endometrial cavity, endometrial sampling may be non-diagnostic.
Treatment includes total abdominal hysterectomy and debulking of tumor if present outside the uterus. Removal of ovaries and lymph nodes remain controversial as metastases to these organs occur in a small percentage and are frequently associated with intra-abdominal disease (Giuntoli). As most leiomyosarcomas are discovered on final pathologic diagnosis following hysterectomies for presumed benign leiomyomata, CT scans are employed to determine if reoperation is necessary for debulking purposes.
Radiation therapy has been shown to decrease local recurrance rates but not improve overall survival. Several different chemotherapy regimens are under investigation.
LMS is generally an aggressive tumor, and even when diagnosed at an early stage. It has a tendency to recur locally and metastasize to the lung and liver. Estimates of 5-year survival range from 45% to 65%.
In a study of 31 patients, the most common recurrence site was lung (5 cases), followed by pelvis and upper abdomen (2 cases); nine patients died of disease with a 5-year overall survival rate of 63% (Kim). Another study of 71 patients concluded that early tumor stage, age <50 years, and absence of vascular space involvement were independently associated with improved prognosis (Mayerhofer).
• Myometrium : Leiomyosarcoma, Epithelioid Variant
Fletcher CDM, ed. Diagnostic Histopathology of Tumors. 3rd Ed. Philadelphia, PA: Elsevier; 2007: 688-692.
Giuntoli RL, et al. Retrospective review of 208 patients with leiomyosarcoma of the uterus: prognostic indicators, surgical management, and adjuvant therapy, Gynecol. Oncol. 89 (2003), 460–469.
Kim WY,et al. Uterine Leiomyosarcoma : 14-year Two-center Experience of 31 Cases. Cancer Res Treat 2009 Mar;41(1):24-8.
Nucci MR, Oliva Esther. Gynecologic Pathology: Foundations in Diagnostic Pathology. Philadelphia, PA: Elsevier: 279-289.
Mahdavi A, et al. Pelvic radiation improves local control after hysterectomy for uterine leiomyosarcoma: a 20-year experience.Int J Gynecol Cancer. 2009 Aug;19(6):1080-4.
Mayerhofer K, et al. Leiomyosarcoma of the uterus: a clinicopathologic multicenter study of 71 cases. Gynecol Oncol 1999 Aug;74(2):196-201.
Mills SE, ed. Sternberg's Diagnostic Surgical Pathology.4th Ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2004: 2510.