
Dilated rounded ducts are lined by epithelial cells that form micropapillary, fenestrated, cribriform, solid with comedo necrosis and Roman-bridge patterns. The neoplastic cells may range from low to high grade. By definition (and analogous to DCIS), the ducts are surrounded by a myoepithelial cell layer. Immunostain with muscle-specific actin would highlight the myoepithelial layer, which may be difficult to visual on routine H&E stain (Fletcher).
A striking similarity to DCIS of the breast can be appreciated. The cysts can be lined by a single of flattened cuboidal cells (center and lower left) or multiple layers of cells with more abundant cytoplasm (see in upper right).
A cribriform pattern is appreciated here with pink hyaline material inside the lumina. The stroma is usually sclerotic.
The tumor cells have eosinophilic or granular cytoplasm with oval or round nuclei with finely dispersed to vesicular chromatin. Nuclei can range from inconspicuous to prominent. A thin myoepithelial layer present at the periphery of the tumor island.
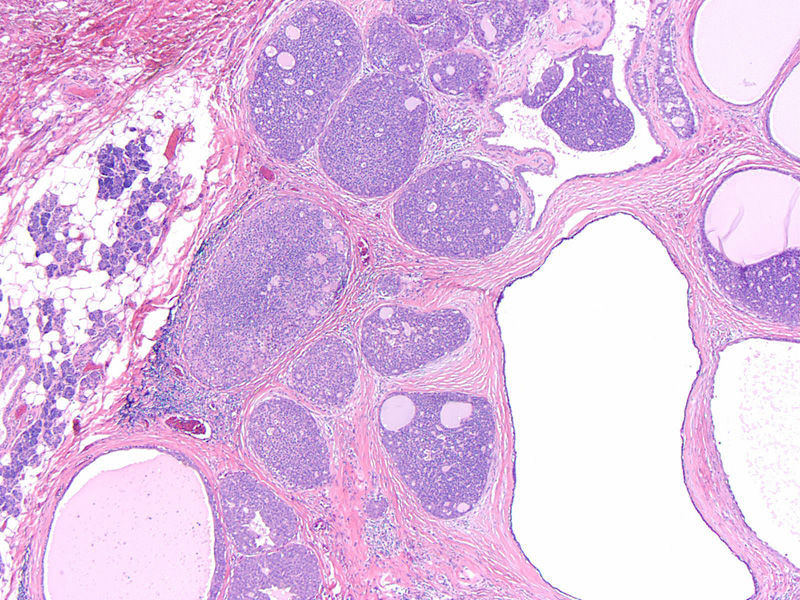
Normal parotid gland is seen on the left and the tumor on the right. Note the irregular infiltration of the neoplasm at the interface and lack of a capsule.
Strong positivity for androgen receptor is seen, which is characteristic of both salivary duct carcinoma and low grade salivary duct carcinoma (McHugh, Simpson)
In 1996, Delgado et al described 10 cases of a low grade-parotid neoplasm with a predominantly intraductal pattern resembling atypical ductal hyperplasia or low grade-intraductal carcinomas of breast (Delgado). Given its resemblance to salivary duct carcinoma but low cytologic grade, these authors proposed the term “low grade salivary duct carcinoma”. They also concluded that this lesion was primarily an in situ process.
Recognition of this entity remains controversial, and the existence of salivary gland malignancies with an exclusive or predominant intraductal component has yet to gain widespread recognition. In fact, the 2005 World Health Organization Classification of Head and Neck Tumors has not included intraductal carcinoma in the classification of salivary gland neoplasms, but do regard this lesion as a variant of cystadenocarcinoma, bestowing the term “low grade cribriform cystadenocarcinoma”.
To add to the increasingly number of appellations, Fletcher prefers the term "intraductal carcinoma". He reasons that if salivary duct carcinomas are analogous to ductal carcinoma of the breast, and low grade salivary duct carcinoma is the in situ counterpart similar to DCIS, then "intraductal carcinoma" emphasizes the in situ and intraductal nature of the neoplasm (Fletcher).
Although only a small number of cases, LG-IDCs seem to have an excellent prognosis. No cases of lymph node metastases, local recurrences or deaths attributed to tumor have been reported in 21 patients with follow-up ranging from 6 months to 11 years (Delgado; Brandwein-Gensler; Chen).
• Salivary Gland : Salivary Duct Carcinoma
• Salivary Gland : Low grade Mucoepidermoid Carcinoma
Brandwein-Gensler M, Hille J, Wang BY et al. Low-grade salivary duct carcinoma: description of 16 cases. Am J Surg Pathol. 2004;28:1040–1044.
Chen KT. Cytology of salivary duct carcinoma. Diagn Cytopathol. 2000;22:132–135.
Delgado R, Klimstra DS, Albores-Saavedra J. Low grade salivary duct carcinoma. A distinctive variant with a low grade histology and a predominant intraductal growth pattern. Cancer. 1996;78:958–967.
McHugh JB, Visscher DW, Barnes EL. Update on selected salivary gland neoplasms. Arch Pathol Lab Med. 2009 Nov;133(11):1763-74.
Simpson RH, Desai S, Di Palma S. Salivary duct carcinoma in situ of the parotid gland. Histopathology. 2008 Oct;53(4):416-25.
Weinreb I, Tabanda-Lichauco R, Van der Kwast T, Perez-Ordoñez B. Low-grade intraductal carcinoma of salivary gland: report of 3 cases with marked apocrine differentiation. Am J Surg Pathol. 2006 Aug;30(8):1014-21.
Images courtesy of and copyrighted by Dr. Jonathan McHugh, Dept of Pathology, University of Michigan.