
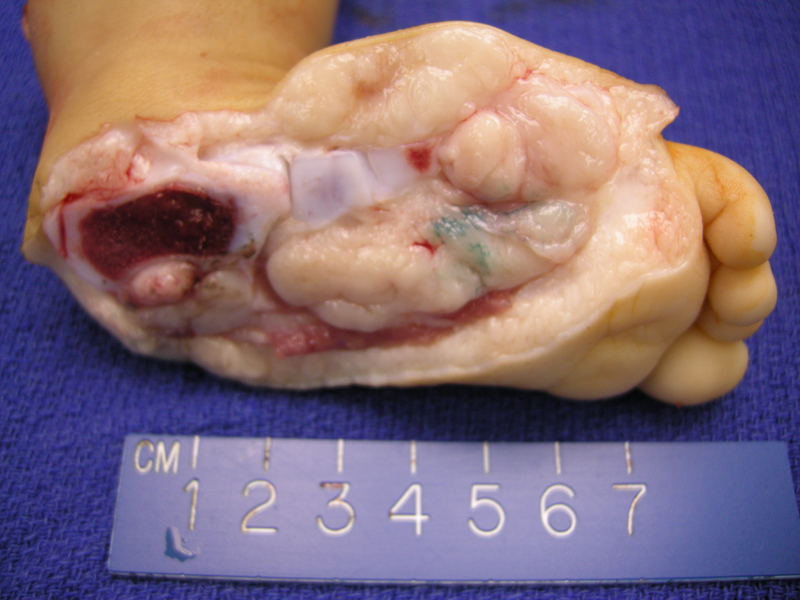
ARMS arising from a child's foot, consisting of a soft fleshy mass in a lobulated configuration. A strip of skeletal muscle can be seen in the lower image.
The classic architecture of ARMS consists of fibrous septae separating tumor cells into clusters. The cells in the center of the nests tend to detach and degenerate, lending a look reminiscent of lung alveoli.
Note the discohesiveness in the center of the cell nests.
Tumor invades adnexal structures (sweat glands seen in upper image).
Neoplastic cells invading the dermis.
Tumors cells single-filing through skeletal muscle.
The neoplastic cells invade through tendon -- longitudinal strips of pink fibers seen most clearly on the right image.
Desmin is an intermediate filament, present in both smooth and striated muscle. Positivity indicates muscle differentiation.3
Myogenin (myogenin gene encodes a protein that induces skeletal muscle differentiation) is strongly positive.3
In general, only the rhabdomyoblasts stain with desmin. Note the cross-striations highlighted by the desmin stain.
This separate case of ARMS demonstrates the typical architecture of clustsers of cells separated by broad bands of connective tissue. Multi-nucleated giant cells with wreath-like formation of nuclei are often described in the literature, but were not identified in this particular case.
Broad fibrous septae separate cords and nests of tumor cells. Note that the center cells are discohesive and detached, creating an open space.
Admixture of rhabdomyoblasts (round eosinophilic cells) with neoplastic undifferentiated (basophilic) cells.
Rhabdomyoblasts can have a spindled morphology. They are often called tadpole or strap cells.
ARMS is associated with a particular translocation between chromosome 2 and 13, creating the PAX3-FOXO1 fusion gene or less frequently, a translocation between chromosome 1 and 13, creating the PAX7-FOXO1 fusion gene. PAX7-FOXO1 tumors have a better prognosis than those harboring the PAX3-FOXO1 mutation.1 Note that FOXO1 is also called the FKHR.
In this FISH image, the combined signals (red and green together creating a yellow signal) indicate a normal chromosome with the target probes in their wildtype configuration. A break-apart signal with green and red signals separated (see nuclei in the top of image) indicates that a translocation has occured.
Tumors of skeletal muscle are usually malignant. Rhabdomyomas (the benign counterpart) are extremely rare. Rhabdomyosarcomas are divided into three main histologic types: embryonal, alveolar and pleomorphic.
In general, the embryonal and alveolar tumors arise in children whereas pleomorphic rhabdomyosarcomas occur in adults. Embryonal lesions tend to affect younger children and the alveolar subtype is more frequent in adolescents. Rhabdomyosarcomas can arise in any anatomic location, but the most common sites are the head and neck, genitourinary tract, limbs and trunk. Alveolar lesions are more likely to arise in the limbs compared to embryonal lesions, which are more common in the head, neck and urogenital regions. Note that in adults, rhabdomyosarcomas are usually located in the limbs, regardless of histologic subtype.1
Grossly, alveolar rhabdomyosarcomas (ARMS) form in the deep musculature of the extremities. Histologically, there is a vague resemblance to alveolar tissue of the lung (hence the name alveolar rhabdomyosarcoma). Fibrous septae separate aggregates of tumor cells. The center of these cell nests degenerate while the periphery cells cling to the fibrous septae, lending the look of pulmonary alveoli. The neoplastic cells are round, with large dark nuclei admixed with rhabdomyoblasts. Rhabdomyoblasts, also know as tadpole or strap cells, are elongated or round cells containing eccentric eosinophilic cytoplasm in which cross-striations may be seen. These cells are immunopositive for muscle markers such as desmin, myogenin and MYOD1.2 Occasionally, multinucleate wreath-like cells are seen.
Note that the solid variant of ARMS can look very much like the embryonal subtype, with diffuse sheets of tumor cells. However, the cells and nuclei of solid ARMS will be larger than embryonal lesions and alveolar nesting will be focally present or highlighted by a reticulin stain. It is important to distinguish between these two subtypes because ARMS is a more aggressive tumor with a poorer prognosis.
Compared to embryonal rhabdomyosarcoma, alveolar rhabdomyosarcoma affects an older age group (10-25) and more commonly arise in the extremities.
In children, overall 5 year survival (for all rhabdomyosarcomas) approaches 70% with a combination of surgery, radiation and chemotherapy. In adults, 5 year survival survival is only ~20%.
Alveolar rhabdomyosarcomas behave more aggressively than embryonal rhabdomyosarcomas and thus, tend to have a poorer outcome. Thus, it is important to distinguish between these two subtypes. Two unique characteristics of alveolar rhabdomyosarcomas (ARMS) include tendency for females to develop breast metastases and to present with extensive bone marrow involvement without evidence of a primary lesions, clinically resembling leukemia.1,3
• Myogenic : Rhabdomyosarcoma, Embryonal Type
1 Fletcher CDM, ed. Diagnostic Histopathology of Tumors. 3rd Ed. Philadelphia, PA: Elsevier; 2007: 1567-1570.
2 Kumar V, Abbas AK, Fausto N. Robbins and Cotran Pathologic Basis of Disease. 7th Ed. Philadelphia, PA: Elsevier; 2005: 1321-2.
3 Rosai, J. Rosai and Ackerman's Surgical Pathology. 9th Ed. Philadelphia, PA: Elsevier; 2004: 2301-4.