
This variant displays a prominent cribriform pattern interspersed with solid squamoid islands (morules).
THe cribriform areas have a swiss cheese appearance, with punctate lumina sometimes arranged back to back.
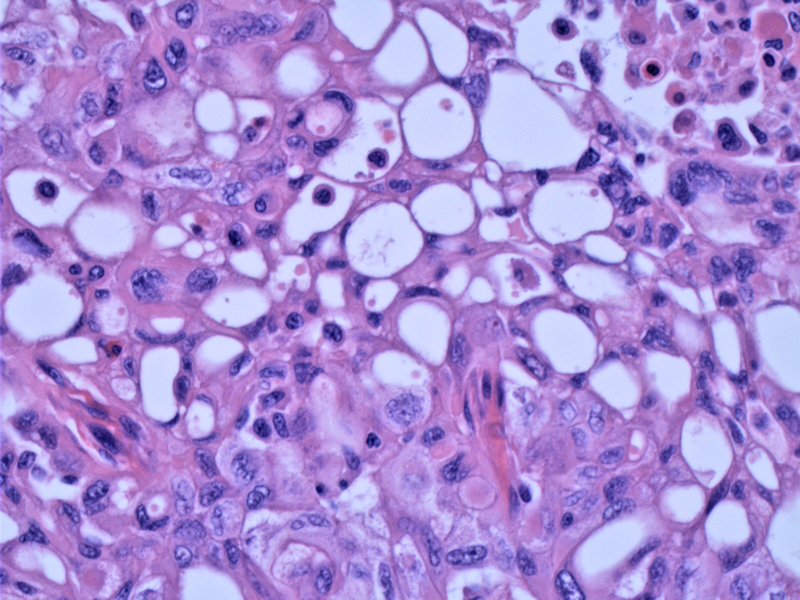
Another area shows solid and cribriform areas.
Another area demonstrating extensive cribriforming. Most of this field consists of lumina rather than cells.
In the solid morular regions, the cells contain abundant eosinophilic to oxyphilic cytoplasm. Note the pink intranuclear inclusion to the right.
This tumor did contain scattered multinucleated giant cells as may be seen in classic PTC.
Intermingling and blending of patterns including more conventional papillary carcinoma is a common feature in this variant.
The tumor elicits marked desmoplasia as it extends into the soft tissues of the neck. Vascular or capsular/extracapsular extension is common in this variant, however, prognosis is similar to classic PTC (Cameselle).
In the lymph node, there is sharply demarcated admixture of cribiform morular and classic patterns of papillary carcinoma. In a study of 9 CMV-PTC, 8 cases demonstrated vascular and capsular invasion and 2 cases demonstrated lymph node metastasis (Cameselle).
Nuclei are focally optically clear with longitudinal grooves and contain eosinophilic, intranuclear inclusions (arrow) as in classic PTC.
Solid squamoid (morular) islands are apparent.
IHC application of beta-catenin in most studies (Hirokawa, Koo) demonstrate both nuclear and membranous staining. Diffuse membranous positivity is demonstrated in this image. There appears to be molecular contributions from the RET/PTC translocations and from mutations in the APC gene and beta-catenin gene.
As would be expected for most thyroid neoplasms, thyroglobulin is positive.
The cribriform-morular variant of papillary thyroid carcinoma (CMV-PTC) is an unusual and peculiar sub-type of papillary thyroid carcinoma. Of interest, this is the variant most commonly seen in familial adenomatous polyposis (FAP) associated PTC. PTC affects 1-2% of patients with familial adenomatous polyposis (FAP) (Alsanea). Patients diagnosed with the cribriform-morular variant of papillary thyroid cancer should be screened for the presence of FAP (Dalal).
The molecular pathogenesis of CMV-PTC is incompletely understood. It appears that there can be molecular contributions from the RET/PTC translocations and from mutations in the APC gene and beta-catenin gene, which are both part of the Wnt signaling pathway. However, one of the most common mutations in papillary carcinoma, the BRAF mutation, has not been reported in this variant of papillary carcinoma (Schuetze).
Commonly arises in young females usually less than 30 years of age. In fact, the female to male ratio is quite remarkable (17:1)(Fletcher). The neoplasms are encapsulated or well-circumscribed in many cases, but extrathyroidal extension and lymph node metastasis can occur, just like in classic PTC (Cameselle). While sporadic forms usually appear as an solitary tumors, those associated with FAP are often multifocal due to different somatic mutations added to the germline mutations (Miyaki).
Thyroid surgery is the main treatment.
Excellent (Cameselle, Perrier). In a study of 9 patients, follow-up was available in seven cases which showed all alive with no evidence of disease at 1 to 13 years after diagnosis, similar to conventional PTC (Cameselle). The 5- and 20-year survival rates of FAP-associated PTCs have been reported to be 90% and 77%, respectively (Perrier).
→The cribriform-morular variant of papillary thyroid carcinoma is the predominant pattern in the setting of FAP.
→The prognosis is similar to that of classic PTC.
• Thyroid : Papillary Carcinoma, Oncocytic Variant
• Thyroid : Papillary Thyroid Carcinoma
• Thyroid : Papillary Carcinoma, Follicular Variant
• Thyroid : Papillary Carcinoma, Solid Variant
• Thyroid : Papillary Carcinoma, Tall Cell Variant
Alsanea O, Clark OH. Familial thyroid cancer. Curr Opin Oncol 2001;13:44–51.
Cameselle-Teijeiro J, Chan JK. Cribriform-morular variant of papillary carcinoma: a distinctive variant representing the sporadic counterpart of familial adenomatous polyposis-associated thyroid carcinoma? Mod Pathol. 1999 Apr;12(4):400-11.
Dalal, K et al. Clinical curiosity: cribriform-morular variant of papillary thyroid carcinoma. Head Neck. 2006 May;28(5):471-6.
Fletcher CDM, ed. Diagnostic Histopathology of Tumors. 3rd Ed. Philadelphia, PA: Elsevier; 2007: 1010.
Hirokawa M, Kuma S, Miyauchi A, et al. Morules in cribriform-morular variant of papillary thyroid carcinoma: immunohistochemical characteristics and distinction from squamous metaplasia. APMIS 2004;112:275–282.
Koo JS, Jung W, Hong SW. Cytologic Characteristics and β-Catenin Immunocytochemistry on Smear Slide of Cribriform-Morular Variant of Papillary Thyroid Carcinoma. Acta Cytol. 2011 Jan-Feb;55(1):13-8. Epub 2010 Nov 26.
Miyaki M. et al. Molecular evidence for multicentric development of thyroid carcinomas in patients with familial adenomatous polyposis. Am J Pathol. 2000;6:1825–1827.
Perrier ND, van Heerden JA, Goellner JR, et al. Thyroid cancer in patients with familial adenomatous polyposis. World J Surg 1998: 22:738–742; discussion 743.
Schuetze D, Hoschar AP, Seethala RR, et al. The T1799A BRAF mutation is absent in cribriform-morular variant of papillary carcinoma. Arch Pathol Lab Med. 2009 May;133(5):803-5.
Author: Lina Aguirre MD, Endocrinology Fellow, University of New Mexico.