
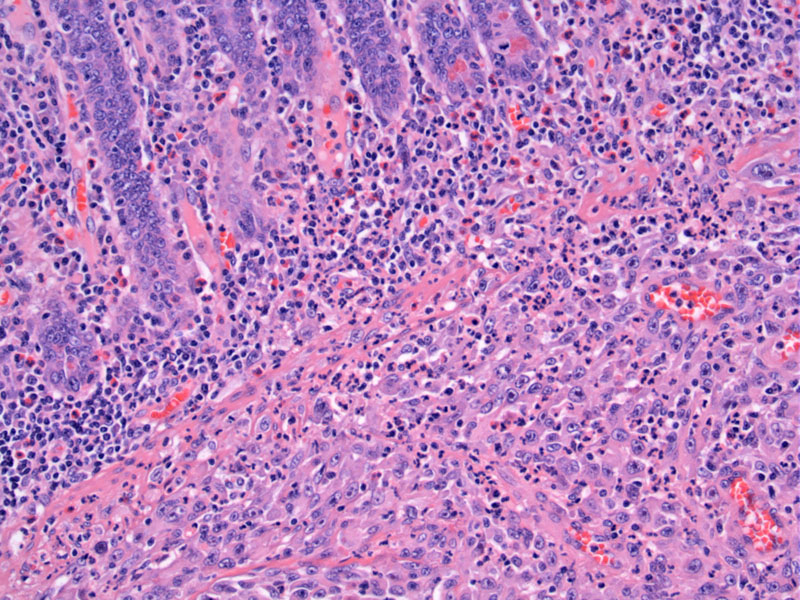
Normal mucosal glands are underminded by a sheet-like growth of malignant epithelioid cells; PDGFRα mutations are frequently associated with epithelioid cell morphology as in this case.
The cells are round with prominent nucleoli, certainly raising the question of poorly differentiated adenocarcinoma. There is an admixed predominantly lymphoid infiltrate.
Multinuceation is common, as shown here by a prominent large cell with at least three round nuclei and eosinophilic cytoplasm. Also other cells appear almost rhaboid looking.
About 8% of GISTs harbor a mutation not in kit but in PDGFRA (platelet derived growth factor receptor alpha). Three different regions of PDGFRA may be mutated: in decreasing order of frequency, exon 18, exon 12, and exon 14. KIT and PDGFRA genes encode highly homologous receptor tyrosine kinase proteins which permit the phosphorylation of the receptor, causing activation of the downstream effectors.
Mutations in PDGFRA arise nearly exclusively in gastric GISTs, mostly in epithelioid variants. Omental GISTs and a few intestinal ones (such as our case) have also been found to harbor this mutation.
Kwon (2009) examined 15 PDGFRA-mutated GISTs located in the stomach and compiled the following unique histopathologic features: (1) epithelioid or mixed type histology; (2) moderate to severe intratumoral infiltration of lymphocytes; (3) presence of rhabdoid appearing cells or multinucleated giant cells; (4) increased pleomorphism; (5) low mitotic activity and (6) low risk of malignancy (Kwon).
These GISTs usually involve the stomach but occasionally intestinal ones are seen.
Extragastric GISTs are thought to be more aggressive than gastric GISTs with the same parameters (Miettinen). Complete resection is the main treatment. Unresectable tumors or those with metastatic disease are treated with Kit/PDGFRA tyrosine kinase inhibitors, such as imatinib.
There is currently much research into KIT-mutated and PGDFRA-mutated GISTs in terms of their behavior. As noted above, the prognosis of GISTs and response to imatinib appears to be dependant on the exact mutation type. For example, gastric GISTs with exon 11 deletions are more aggressive than those with substitutions (Lasota).
A review of 289 PGDFRA-mutated GISTs, Corless (2005) found that two-thirds exhibited mutations in exon 18 (specifically the D842V substitution) and are resistant to imatinib (Corless). Missense mutations in exon 14 seem to be associated with low Kit expression and unexpectedly favorable prognosis (Lasota).
• Small Intestines : Gastrointestinal Stromal Tumor (Duodenum)
• Stomach : Gastrointestinal Stromal Tumor (GIST)
Corless, et al. PDGFRA mutations in gastrointestinal stromal tumors: frequency, spectrum and in vitro sensitivity to imatinib.. J Clin Oncol 2005. 23:5357–5364.
Hou YY, et al. Impact of KIT and PDGFRA gene mutations on prognosis of patients with gastrointestinal stromal tumors after complete primary tumor resection. J Gastrointest Surg. 2009 Sep;13(9):1583-92.
Kwon JE, et al. Pathological characteristics of gastrointestinal stromal tumours with PDGFRA mutations. Pathology. 2009;41(6):544-54.
Lasota, J, Stachura J, and Miettinen M. GISTs with PDGFRA exon 14 mutations represent subset of clinically favorable gastric tumors with epithelioid morphology. Lab Invest 2006. 86:94–100.
Lasota J, Miettinen M. Clinical significance of oncogenic KIT and PDGFRA mutations in gastrointestinal stromal tumours. Histopathology. 2008 Sep;53(3):245-66.
Miettinen M, Lasota J. Gastrointestinal stromal tumors: review on morphology, molecular pathology, prognosis, and differential diagnosis. Arch Pathol Lab Med. 2006 Oct;130(10):1466-78.