
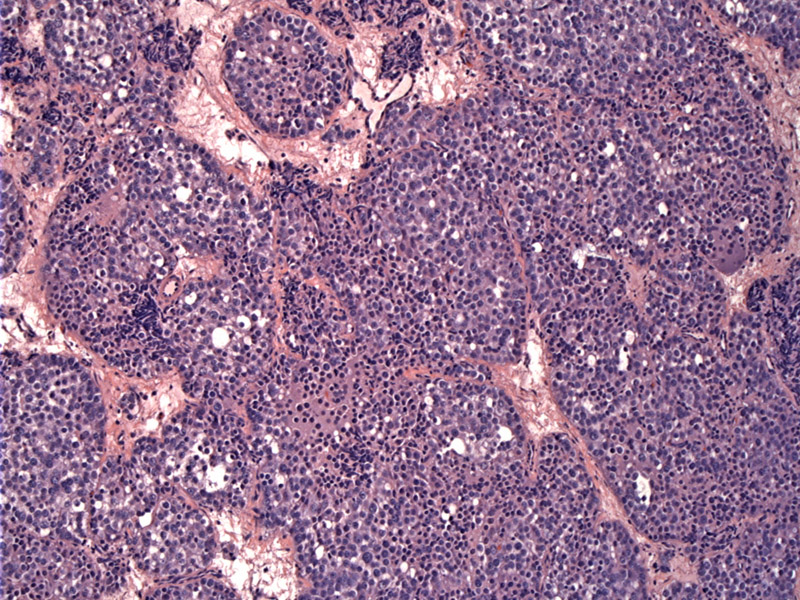
Noncohesive cells with very scant intervening stroma comprises spermatocytic seminoma. There is also no glycogen, so cytoplasmic clearing seen in classic seminoma is lacking. Mitoses including abnormal forms are frequent, as seen here. Note also the lack of intervening lymphocytes, which are so prominent in classic seminoma.
There are typically three cell types, with the predominant cell type of medium size with a variable amount of dense eosinophilic cytoplasm and a round nucleus, often with a fine granular chromatin. The second type of cell is smaller with dark-staining nuclei and scant eosinophilic cytoplasm resembling a lymphocyte. The third cell type is large mononucleated or, rarely, multinucleated with round, oval, or indented nuclei. The cell type may appear in many areas to be relatively monotonous, although wider sampling reveals characteristic areas.
The tumor is divided into lobules which are closely approximated and devoid of intervening lymphocytes.
Spermatocytic seminomas are distinct in their clinical, pathological, and genetic natures from classic seminoma. They are most likely to be confused with lymphoma and classic seminoma, but familiarity with their unique cell types should avoid misclassification. Unlike classic seminoma, for example, these tumors are not associated with intratubular germ cell neoplasia. Additionally, the gain of chromosome 9 is a consistent finding in all spermatocytic seminomas, and is not found in classic seminomas (Verdorfer).
Grossly, spermatocytic seminomas tend to be larger than classic seminomas and also, more likely to be bilateral and multifocal. Microscopically, these tumors are defined by the presence of three cell types: (1) medium-sized cells between 15-18 nanometers with round nuclei containing fine chromatin and eosinophilic cytoplasm; (2) small dense cells between 6-8 nanometers; (3) giant cells between 50-100 nanometers that may be uninucleate or multinucleate. The stroma may be quite edematous.1,2
Affects older men compared to most testicular tumors. Median age of onset is 52, which is 12 years later than usual seminoma.1 In contrast to classic seminomas, they present as slow-growing painless masses that rarely (if ever) metastasize.2
Orchiectomy alone without any form of adjuvant therapy is considered adequate.
Excellent; pure spermatocytic seminomas lack the capacity to metastasize (Talerman). Note, however, that spermatocytic seminomas with sarcomatous change have a grim prognosis with death within a year, however, sarcomatous change is exceptionally rare and only 12 cases have been reported thus far.1
• Testis : Seminoma, Classic Type
• Testis : Seminoma, Classic Type
1 Zhou M, Magi-Galluzzi, C. Genitourinary Pathology: Foundations in Diagnostic Pathology. Philadelphia, PA: Elvesier; 2006: 544-8.
2 Kumar V, Abbas AK, Fausto N. Robbins and Cotran Pathologic Basis of Disease. 7th Ed. Philadelphia, PA: Elsevier; 2005: 1042-3.
Aggarwal N, Parwani AV. Spermatocytic seminoma. Arch Pathol Lab Med. 2009 Dec;133(12):1985-8.
Talerman, A. Spermatocytic seminoma: clinicopathological study of 22 cases. Cancer 1980. 45 8:2169–2176.
Verdorfer, I. , H. Rogatsch , A. Tzankov , H. Steiner , and G. Mikuz . Molecular cytogenetic analysis of human spermatocytic seminomas. J Pathol 2004. 204 3:277–281.