
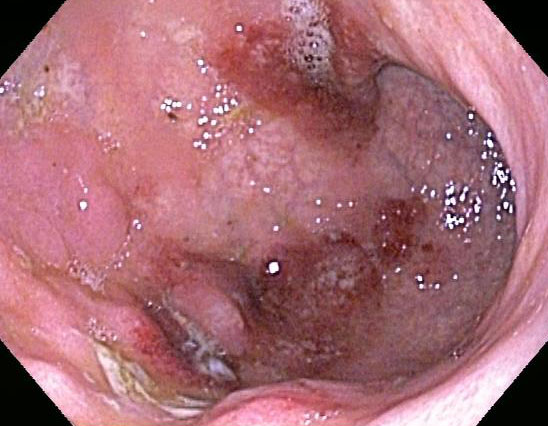
Endoscopy shows a diffuse polypoid lesion with coarse mucosal granularity.
Most lymphomas are limited to the mucosa or submucosa as seen here. This submucosal infiltrate of small blue cells devoid of architecture produces a polypoid mass. Most lymphomas are superficial, but those that show deeper wall extension may be associated with lymph node involvement.
Reactive gastric glands with intestinal metaplasia remain but the normal mucosa is otherwise effaced secondary to a dense lymphoid infiltrate, with some germinal centers.
The tumor consists of a diffuse proliferation of monotonous lymphocytes with a few plasma cells and eosinophils admixed.
The malignant lymphoid cells are small to medium-sized with slightly irregular nuclei harboring a centrocyte appearance. Sometimes reactive lymphoid follicles can be discerned which may be vaguely present here.
Mitoses are also noted.
There is a monomorphic infiltrate of lymphoid cells with a rim of clear cytoplasm.
CD20 is strongly positive and highlights the malignant cell population.
By comparison, CD3 highlights reactive T cells within the infiltrate.
Extranodal lymphoma (i.e. lymphomas that arise in an organ or tissue other than a lymph node or spleen) frequently involves in the GI tract. Within the GI tract, the stomach is the most common site (60-75% of cases) followed by the small bowel and large bowel (Psyrri).
Lymphomas in the stomach are, for the most part, of B-cell lineage. There are two major subtypes: (1) those that arise from mucosa-associated lymphoid tissue, so called MALT lymphomas or MALTomas; (2) diffuse large B-cell lymphoma (DLBCL), which can occur de novo or arise from a pre-existing MALT lymphoma (Psyrri).
There is compelling evidence that H. pylori is the underlying causative agent of gastric MALT lymphoma. Over 90% of these lesions arise from chronic gastritis induced by Helicobacter infection (Fletcher). The role of H. pylori infection in the genesis of DLBCL, however, is unclear.
Three different translocations have been identified in a subset (~65%) of gastric MALT (Ferrucci). Although these translocations involve fusion of different genes, all three mutations converge to constitutively activate the oncogenic pathway involving nuclear factor NFkB:
The t(11;18) translocation is the most frequent chromosome aberration, seen in approximately 1/3 of MALT lymphomas (Ferrucci) and involves fusion of the API2 and the MALT1 gene.
Gastric MALT lymphomas bearing this mutation fail to respond to H. pylori therapy and these patients are more likely to have disseminated disease. On the bright side, this mutation is not seen in MALT lymphomas with a diffuse large B-cell component, and thus, may confer "immunity" or resistance against this transformation (Fletcher, Ferrucci).
The t(14;18) translocation juxtaposes the MALT1 gene with the IGH gene, and is seen in about 20% of MALT lymphomas. This translocation is more common in the salivary glands, eye, skin and lung (Fletcher).
The t(1;14) translocation joins the bcl-10 gene to the IGH gene, leading to overexpression of bcl-10 protein. This translocation only occurs in 1-2% of cases and is associated a more aggressive tumor (Fletcher); and like the t(11;18) translocation, are less like to respond to H. pylori eradication therapy.
The tumor itself is a low-grade B-cell lymphoma and has the following immunoprofile: positive for CD19/CD20 (B-cell markers) as well as bcl2. It is negative for CD5/CD10/CD23/cyclin D1/bcl-6. Co-expression of CD43 is seen in approximately 50% of cases (Fletcher).
Symptoms often mimic gastritis and include epigastric pain and dyspepsia. Shallow ulcers or mucosal granularity may be found endoscopically.
H. pylori-positive patients with stage I can undergo treatment of H. Pylori eradication. If H. Pylori infection persists, re-eradication should be attempted. Endoscopy and biopsies every 3–6 months is recommended. If complete regression of macroscopic disease is not attained after 12 months, radiation alone or rituximab or single-agent chemotherapy if RT is contraindicated (Psyrri). Those with macroscopic remission but minimal lymphoma histologically may undergo watchful waiting with regular biopsies (every 3 months) (Psyrri).
H. pylori-negative patients, or those with stage II or/and with t(11;18) translocation may receive antibiotic treatment in addition to close follow-up with endoscopy every 3 months. Combined chemotherapy alone or plus radiation should be initiated if no regression is seen.
For those occasional cases presenting with higher stage, chemotherapy is the treatment of choice in most cases.
Overall 5 year survival exceeds 90%. Approximately 10% of gastric MALT lymphomas are unresponsive to Helicobacter pylori eradication. The API2-MALT1 fusion from chromosome translocation t(11;18)(q21;q21) has been reported as a predictor of non-responsiveness to H. pylori eradication therapy (Ferruci). This particular translocation is also associated with multifocality (Fletcher).
Despite the growing body of evidence that up to 25% percentage of patients with gastric MALT lymphomas have multiorgan disease, there is no significant difference in survival between those with localized and those with disseminated disease (Fletcher).
A small percentage of tumors progress to diffuse large B-cell lymphoma.
The t(11;18) translocation and/or translocation t(1;14)(p22;q32) identifies cases that do not respond to H. Pylori eradication.
• Salivary Gland : MALT Lymphoma
• Salivary Gland : Extranodal Marginal Zone Lymphoma (of parotid)
• Stomach : H. pylori Gastritis
Fletcher CDM, ed. Diagnostic Histopathology of Tumors. 3rd Ed. Philadelphia, PA: Elsevier; 2007: 1193-6.
Ferrucci PF, Zucca E. Primary gastric lymphoma pathogenesis and treatment: what has changed over the past 10 years? Br J Haematol. 2007 Feb;136(4):521-38. Epub 2006 Dec 8.
Psyrri A, Papageorgiou S, Economopoulos T. Primary extranodal lymphomas of stomach: clinical presentation, diagnostic pitfalls and management. Ann Oncol. 2008 Dec;19(12):1992-9.
