
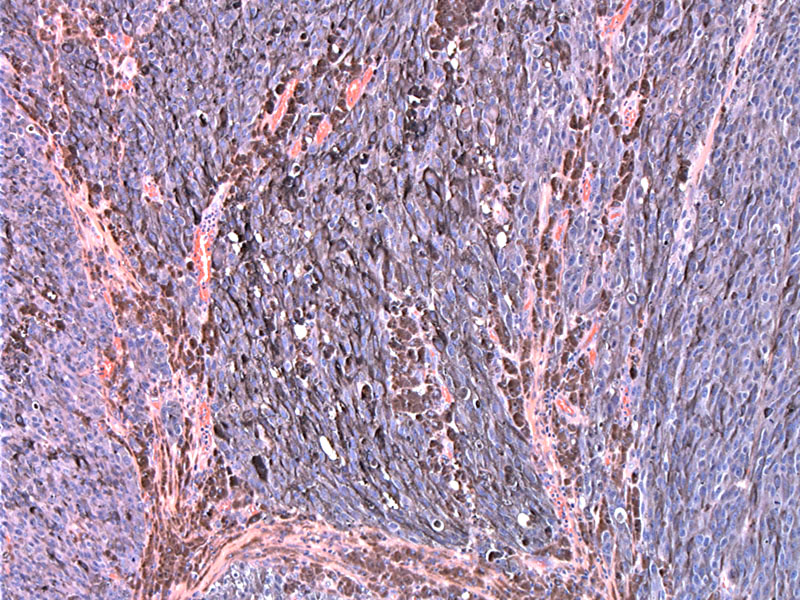
Nodules of heavily pigmented cells, sometimes obscuring the cytological features, penetrate deep into the dermis.
The overlying surface is undermined by the malignant proliferation of dermal melanocytes.
Spindled melanocytes are seen in the dermal nodule.
Other areas demonstrate a more epithelioid morphology. While this case shows features typical of melanoma, this tumor is a great mimicker, including some pitfalls with inmmunohistochemistry. Remember that cytokeratin, a specific marker for epithelial differentiation, has been reported in 10% to 26% of metastatic melanomas (Banks), although it is far less prevelant in primary cutaneous melanoma. Also, melanomas with small cell morphology and aberrant expression of neuroendocrine markers exist, and these tumors may be only focally positive or negative for S100 (Eyden).
The malignant melanocytes lack cohesion, show marked nuclear pleomorphism and eccentric nuclei. These features are typical of most variants of melanoma.
Case 2: Typically, the nodular variant does not demonstrate an intrepithelial (in situ) component as seen here.
Note the dermal proliferation of epithelioid melanocytes with prominent nucleoli.
The nested malignant melanocytes in the dermis are obviously pleomorphic.
The four most common variants of melanoma are the superficial spreading type, lentigo maligna type, nodular type and acral type.
The nodular subtype exhibits little radial growth and predominantly vertical growth. Clinically, this type forms a blue/black nodule or elevated plaque, or as a polypoid, sometimes ulcerated tumor. There is no surrounding flat pigmented lesion seen in the nodular type (a common finding in the superficial spreading or lengito maligna type). Histologically, there is a dermal proliferation of melanocytes. The intraepidermal component is either entirely absent or minimal (Duncan).
Nodular melanomas have a tendency to affect slightly older patients than superficial spreading melanoma (median age: 7th decade). They arise at any location, and may be clinically confused with other cutaneous tumours such as basal cell carcinomas, particularly if they are amelanotic.
Lymph node biopsy is recommended for patients with intermediate-thickness melanomas (Breslow thickness, 1-4 mm).
Due to its rapid growth and predominantly vertical invasive growth, the nodular variant is considered the most aggressive of the four conventional subtypes. Prognosis is influenced by histologic parameters, such as tumor thickness, mitoses, tumor regression, tumor-infiltrating lymphocytes, ulceration, satellitosis, and angiolymphatic spread. When the regional nodes are positive, 5-year survival rate decreases to approximately 30% to 40%, with worsening of prognosis with increasing number of involved lymph nodes.
• Melanocytic : Melanoma, Superficial Spreading Type
• Neuroendocrine : Merkel Cell Carcinoma
• Uncertain Lineage : Clear Cell Sarcoma
Banks ER, Jansen JF, Oberle E, Davey DD . Cytokeratin positivity in fine-needle aspirates of melanomas and sarcomas. Diagn Cytopathol. 1995;12(3):230–233.
Duncan LM. The classification of cutaneous melanoma. Hematol Oncol Clin North Am. 2009 Jun;23(3):501-13, ix.
Eyden B, Pandit D, Banerjee SS . Malignant melanoma with neuroendocrine differentiation: clinical, histological, immunohistochemical and ultrastructural features of three cases. Histopathology. 2005;47(4):402–409
Pollack LA, et al. Melanoma survival in the United States, 1992 to 2005. J Am Acad Dermatol. 2011 Nov;65(5 Suppl 1):S78-86.