
Case 1: A proliferation of epithelioid-appearing cells and some spindled cells comprise this MPNST. MPNSTs composed of predominantly epithelioid cells are usually nested and the cells can even appear rhabdoid with macronucleoli (Folpe).
Mitotic figures are usually easy to find. Here, the cells are more spindled in areas.
Case 2 - Myxoid MPNST: about 10% of MPNST have abundant myxoid stroma.
There is a cellular proliferation of cells with round nuclei in a myxoid stroma.
Vasculocentric proliferation of tumor cells is a characteristic feature and is mostly present in this field.
Patchy S100 staining occurs in spindled MPNST whereas the epithelioid variant (not shown) generally exhibits diffuse S100 positivity.
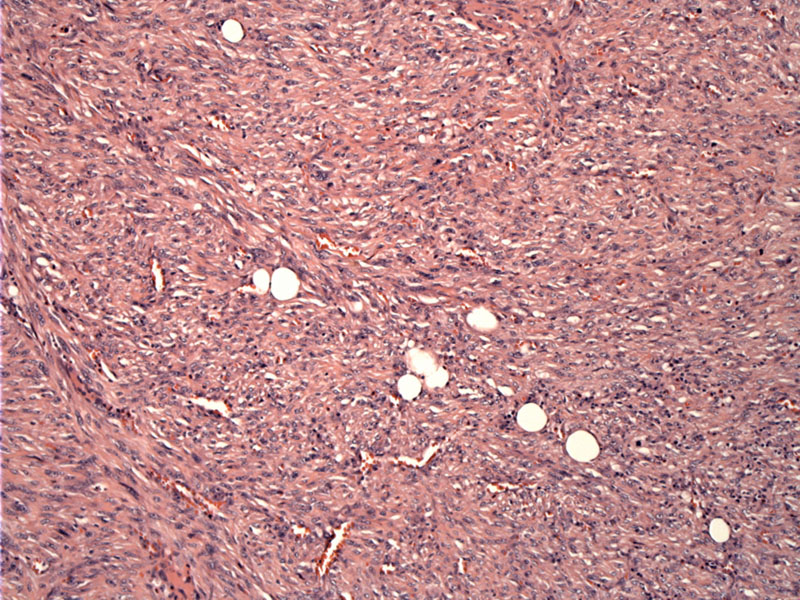
Case 3: This tumor is typical in morphology with its fibrosarcomatous growth pattern. Fascicles intersect at varying angles and a focal herringbone pattern is discernable.
Ectatic thin walled vessels as can be seen in hemangiopericytoma may be encountered in this tumor. Tumor cells show diffuse hyperchromasia.
MPNSTs can arise de novo or from an existing neurofibroma in a patient with NF1. Approximately half of all MPNSTs are NF1-associated (Folpe, Fletcher). Neurofibromatosis 1 ("von Recklinghausen disease") is an autosomal dominant genetic disorder caused by a mutation of neurofibromin on chromosome 17q. Lesions seen in NF1 include neurofibromas, cafe-au-lait spots, optic gliomas, Lisch nodules, skeletal deformities. Recall that NF2 is a mutation of the gene merlin on chromosome 22. Lesions seen in NF2 include bilateral acoustic neuromas and increased risk of ependymomas and meningiomas.
Grossly, a fusiform tumor is seen attached to a nerve or arising from a pre-existing neurofibroma. The tumor is firm or hard, but usually exhibits areas of necrosis and hemorrhage. Histologically, fascicular or storiform proliferations of spindled (95%) or epithelioid (5%) cells are seen. Alternating zones of cellularity impart a marbled appearance in some cases, but others are uniformly cellular. There is accentuated perivascular cellularity -- protrusion of tumor into the vessels is particularly characteristic and may lead to thrombosis of the vessel.
Of interest, epithelioid and spindled MPNSTs have different IHC profiles. For example, spindled MPNSTs are only focally positive for S-100 whereas epithelioid MPNSTs are diffusely positive.
A peculiar variant called "malignant Triton tumor" consists of rhabdomyoblasts, mucinous glands and metaplastic osteoid/cartilege and are seen in NF1 patients (Folpe). MPNSTs with rhabdomyoblastic differentiation will stain for muscle markers (desmin, myogenin, MyoD1)(Folpe).
Monophasic synovial sarcoma (mSS), in particular, can really mimic MPNST, but wiry collagen seen in synovial sarcoma is usually absent in MPNST. IHC will be necessary to sort out some cases. For example, MSS is generally positive for cytokeratins whereas MPNST is negative. mSS also exhibit the t(x;18) SYT-SSX translocation.
De novo tumors tend to arise in the fourth decade whereas NF1-associated tumors arise earlier. It is important to consider NF1 if an adolescent patient presents with a MPNST. The lifetime risk for a NF1 patient is 2% (Fletcher).
There is a male predominance (in NF1 associated tumors) and the most common anatomic locations include the buttocks, thigh, upper arm and paraspinal region (Folpe). These typically are deep-seated tumors.
Resection of the tumor is indicated with negative margins. Often post operative radiation is also employed. Chemotherapy is only used in cases in which the chance of metastatic disease is high.
Local recurrences are not uncommon, with rates in the range of 40-65%; the rate for distant metastases is similar, with a predilection for pulmonary metastasis, followed by bone then pleura. Overall 5 year survival is about 50% in sporadic cases and 25% in NF1 patients (Fletcher). Better outcomes are seen with small tumors, low grade tumors, and those excised with negative margins.
→Can arise de novo or in the setting of NF1
→Spindled MPNST is most common. Myxoid, epithelioid and rhabdomyoblastic differentiation are distinct subtypes.
→Perivascular accentuation of tumor cells is a particular characteristic feature.
• Uncertain Lineage : Clear Cell Sarcoma
Fletcher CDM, ed. Diagnostic Histopathology of Tumors. 3rd Ed. Philadelphia, PA: Elsevier; 2007: 1747-1750.
Folpe AL, Inwards CY. Bone and Soft Tissue Pathology: Foundations in Diagnostic Pathology Philadelphia, PA: Elsevier; 2010: 214-9.