
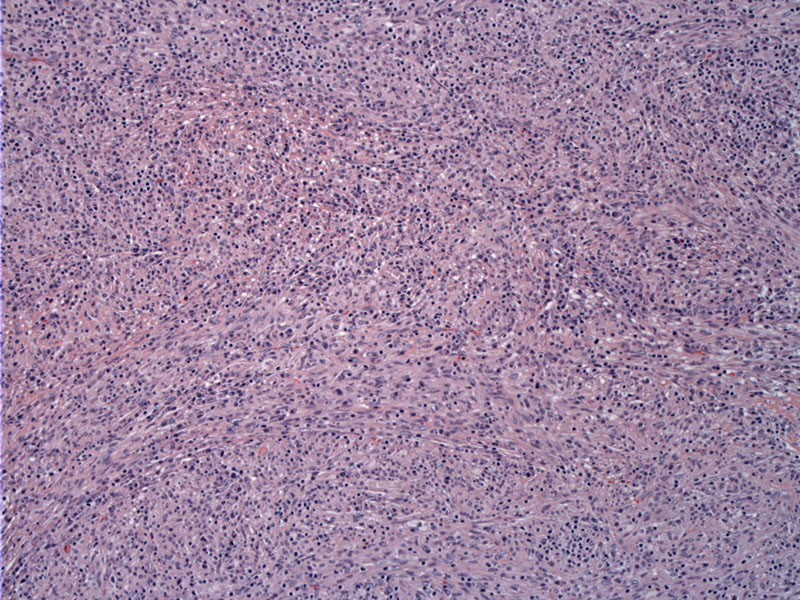
Low power shows replacement of liver parenchyma by a spindled eosinophilic cellular process with many inflammatory cells. There is some vague fascicular growth, but overall the pattern is rather disordered.
A solid mass is seen composed of spindled cells admixed with a sprinkled inflammatory cells.
Plasma cells predominate as the inflammatory component. Lymphocytes are also seen as well as occasional eosinophils.
Many inflammatory cells do stain for CD68, a macrophage marker.
Smooth muscle actin (SMA) highlights some of the spindle cells. In general, IMTs are uniformly positive for vimentin, and the majority of cases show spindle cells reactive for smooth muscle actin and/or muscle-specific actin, supporting the myofibroblastic nature of these cells. There is no reactivity for myogenin, myoglobin, CD34, CD117, or S100. Reactivity for desmin and cytokeratin may be encountered.
Inflammatory myofibroblastic tumor (IMT) is a rare neoplasm that is usually identified in the lungs and abdomen (mesentery or omentum) of children and young adults (Solomon). Visceral involvement is distinctly uncommon, but about 80 cases of IMT in the liver have been reported in the literature (Schnelldorfer).
The exact nature of IMT is unclear. There is evidence that it is a reactive process as some IMTs occur after trauma, surgery or infection, however, recent molecular studies have found translocations involving the ALK gene in more than half of IMTs (Solomon). ALK gene rearrangement has been documented with a variety of fusion partners––ATIC, CARS, TPM3, TMP4, TPM3 and TPM4, CLTC, RANBP2, and SEC31L– specified in fewer than 30 cases. The influence of ALK expression on clinical behavior is inconclusive, with no known prognostic significance among the different genetic fusions.
Histologically, IMTs (at any site) are characterized by a spindled myofibroblasts in fascicles with scattered inflammatory cells with frequent plasma cells (hence the lesions older name-- plasma cell granuloma.
Similar to IMTs in other sites, patients may exhibit constitutional symptoms such fever, fatigue and weight loss. Laboratory abnormalities include leukocytosis, anemia, thrombocytosis and elevated ESR (Solomon). Specific to liver IMTs, the patient may exhibit right upper quadrant pain and hepatomegaly. IMT of the liver tends to be located in the periphery and only a handful of cases in the hilum have been reported (Schelldorfer).
Surgical resection is the treatment of choice whenever possible. Note that regression and response to corticosteroids and nonsteroidal anti-inflammatory drugs has been noted.
IMT may mimic malignany but usually following complete surgical resection, these tumors generally behave in an indolent manner (Solomon). Of course, rare cases with local recurrence and aggressive clinical course have been reported. The recurrence rate appears related to factors that preclude complete excision, such as inaccessible site, proximity to vital structures, and multifocal
lesions (Solomon).
• Bladder : Pseudosarcomatous Myofibroblastic Proliferation
• Bladder : Urachal Inflammatory Myofibroblastic Tumor
• Peritoneum : Inflammatory Myofibroblastic Tumor
• Spleen : Inflammatory Pseudotumor
• Lung : Inflammatory Myofibroblastic Tumor
Schnelldorfer T, Chavin KD, Lin A et al. Inflammatory Myofibroblastic Tumor of the Liver. J Hepatobiliary Pancreat Surg (2007); 14:421-423.
Solomon GJ, Kinkhabwala MM, Akhtar M. Inflammatory Myofibroblastic Tumor of the Liver. Arch Pathol Lab Med. 2006;130:1548-1551.
Chen ST, Lee JC. An inflammatory myofibroblastic tumor in liver with ALK and RANBP2 gene rearrangement: combination of distinct morphologic, immunohistochemical, and genetic features. Hum Pathol. 2008 Dec;39(12):1854-8. Epub 2008 Aug 12.